Pridopidine
Pridopidine (formerly Huntexil, ACR16, also ASP2314) is an orally bioavailable small molecule investigational drug candidate. It is a highly selective Sigma-1 Receptor (S1R) agonist.[1][2] The S1R regulates key cellular processes relevant to neurodegenerative diseases, such as calcium homeostasis, cytoskeleton dynamics, restoring mitochondrial health and neurotrophic factor release. S1R is implicated in cellular differentiation, neuroplasticity, neuroprotection, and cognitive functioning of the brain.
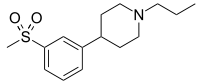 | |
| Names | |
|---|---|
| IUPAC name
4-(3-(Methylsulfonyl)phenyl)-1-propylpiperidine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.240.998 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H23NO2S | |
| Molar mass | 281.41 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Pridopidine positively influences S1R regulated pathways across neurodegenerative and neurodevelopmental indications, including protection against axonal and neuronal injury, restoring spine impairments, enhancing BDNF secretion and restoring mitochondrial function.
Priodopine was previously thought to be a dopamine D2 receptor (D2R) antagonist but this was discovered to be incorrect.[3]
Pridopidine is in late-stage development by Prilenia Therapeutics. Previously it was owned by Teva Pharmaceutical Industries who acquired the rights to the product from its original developer NeuroSearch in 2012.
Pridopidine is the first drug to show statistically significant effect on maintenance of functional capacity in Huntington's Disease (HD),[4] as measured by Total Functional Capacity (TFC). This effect was most prominent in early HD patients.[5]
While Pridopidine at low doses affects S1R, at higher doses it interacts with other targets including dopamine D3, adrenergic α2C and serotoninergic 5-HT1A, which are established therapeutic targets for Parkinson's Disease Levodopa Induced Dyskinesia (PD-LID). This is the likely mechanism for the clear effect seen in the “gold standard” non-human primate model for PD-LID – the MPTP-Lesioned Macaques.[6] This model is considered highly translatable to success in phase 2 clinical trials.
Pridopidine is currently in phase 2 clinical development for PD-LID,[7] and was recently chosen[8][9] to participate in a novel platform trial for amyotrophic lateral sclerosis (ALS) by the Sean M. Healey & AMG Center for ALS at Massachusetts General Hospital.
References
- Johnston, Tom H.; Geva, Michal; Steiner, Lilach; Orbach, Aric; Papapetropoulos, Spyros; Savola, Juha-Matti; Reynolds, Ian J.; Ravenscroft, Paula; Hill, Michael (May 2019). "Pridopidine, a clinic-ready compound, reduces 3,4-dihydroxyphenylalanine-induced dyskinesia in Parkinsonian macaques". Movement Disorders. 34 (5): 708–716. doi:10.1002/mds.27565. ISSN 1531-8257. PMID 30575996.
- Sahlholm, Kristoffer; Sijbesma, Jurgen W. A.; Maas, Bram; Kwizera, Chantal; Marcellino, Daniel; Ramakrishnan, Nisha K.; Dierckx, Rudi A. J. O.; Elsinga, Philip H.; van Waarde, Aren (September 2015). "Pridopidine selectively occupies sigma-1 rather than dopamine D2 receptors at behaviorally active doses". Psychopharmacology. 232 (18): 3443–3453. doi:10.1007/s00213-015-3997-8. ISSN 1432-2072. PMC 4537502. PMID 26159455.
- Sahlholm, Kristoffer; Sijbesma, Jurgen W. A.; Maas, Bram; Kwizera, Chantal; Marcellino, Daniel; Ramakrishnan, Nisha K.; Dierckx, Rudi A. J. O.; Elsinga, Philip H.; van Waarde, Aren (September 2015). "Pridopidine selectively occupies sigma-1 rather than dopamine D2 receptors at behaviorally active doses". Psychopharmacology. 232 (18): 3443–3453. doi:10.1007/s00213-015-3997-8. ISSN 1432-2072. PMC 4537502. PMID 26159455.
- "Huntington's Patients May See Delay in Functional Decline with Pridopidine". Huntington's Disease News. 2017-04-20. Retrieved 2019-09-21.
- "Trial finds Teva drug could slow Huntington's disease". en.globes.co.il (in Hebrew). 2016-09-19. Retrieved 2019-09-21.
- Johnston, Tom H.; Geva, Michal; Steiner, Lilach; Orbach, Aric; Papapetropoulos, Spyros; Savola, Juha-Matti; Reynolds, Ian J.; Ravenscroft, Paula; Hill, Michael (May 2019). "Pridopidine, a clinic-ready compound, reduces 3,4-dihydroxyphenylalanine-induced dyskinesia in Parkinsonian macaques". Movement Disorders. 34 (5): 708–716. doi:10.1002/mds.27565. ISSN 1531-8257. PMID 30575996.
- "A Study to Assess the Safety and Effectiveness of Pridopidine Compared to Placebo in the Treatment of Levodopa-Induced Dyskinesia in Patients With Parkinson's Disease - Full Text View - ClinicalTrials.gov". clinicaltrials.gov. Retrieved 2019-09-21.
- "Sean M. Healey & AMG Center for ALS at Mass General launches first ALS Platform Trial with 5 promising drugs". Massachusetts General Hospital. Retrieved 2019-09-21.
- "Prilenia's Pridopidine Chosen to Participate in the First ALS Platform Trial". www.businesswire.com. 2019-09-18. Retrieved 2019-09-21.