Kinase
In biochemistry, a kinase is an enzyme that catalyzes the transfer of phosphate groups from high-energy, phosphate-donating molecules to specific substrates. This process is known as phosphorylation, where the substrate gains a phosphate group and the high-energy ATP molecule donates a phosphate group. This transesterification produces a phosphorylated substrate and ADP. Conversely, it is referred to as dephosphorylation when the phosphorylated substrate donates a phosphate group and ADP gains a phosphate group (producing a dephosphorylated substrate and the high energy molecule of ATP). These two processes, phosphorylation and dephosphorylation, occur four times during glycolysis.[2][3][4]
Kinases are part of the larger family of phosphotransferases. Kinases should not be confused with phosphorylases, which catalyze the addition of inorganic phosphate groups to an acceptor, nor with phosphatases, which remove phosphate groups (dephosphorylation). The phosphorylation state of a molecule, whether it be a protein, lipid or carbohydrate, can affect its activity, reactivity and its ability to bind other molecules. Therefore, kinases are critical in metabolism, cell signalling, protein regulation, cellular transport, secretory processes and many other cellular pathways, which makes them very important to human physiology.
Biochemistry and functional relevance
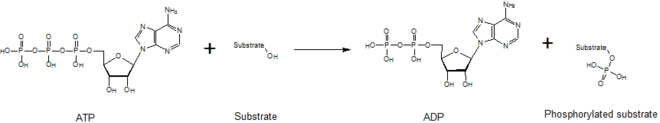
Kinases mediate the transfer of a phosphate moiety from a high energy molecule (such as ATP) to their substrate molecule, as seen in the figure below. Kinases are needed to stabilize this reaction because the phosphoanhydride bond contains a high level of energy. Kinases properly orient their substrate and the phosphoryl group within their active sites, which increases the rate of the reaction. Additionally, they commonly use positively charged amino acid residues, which electrostatically stabilize the transition state by interacting with the negatively charged phosphate groups. Alternatively, some kinases utilize bound metal cofactors in their active sites to coordinate the phosphate groups. Protein kinases can be classed as catalytically active (canonical) or as pseudokinases, reflecting the evolutionary loss of one or more of the catalytic amino acids that position or hydrolyse ATP.[5] However, in terms of signalling outputs and disease relevance, both kinases and pseudokinases are important signalling modulators in human cells, making kinases very important drug targets.[6]
Kinases are used extensively to transmit signals and regulate complex processes in cells. Phosphorylation of molecules can enhance or inhibit their activity and modulate their ability to interact with other molecules. The addition and removal of phosphoryl groups provides the cell with a means of control because various kinases can respond to different conditions or signals. Mutations in kinases that lead to a loss-of-function or gain-of-function can cause cancer[7] and disease in humans, including certain types of leukemia and neuroblastomas, glioblastoma,[8] spinocerebellar ataxia (type 14), forms of agammaglobulinaemia, and many others.[9]
History and classification
The first protein to be recognized as catalyzing the phosphorylation of another protein using ATP was observed in 1954 by Gene Kennedy at which time he described a liver enzyme that catalyzed the phosphorylation of casein. In 1956, Edmond H. Fischer and Edwin G. Krebs discovered that the interconversion between phosphorylase a and phosphorylase b was mediated by phosophorylation and dephosphorylation.[10] The kinase that transferred a phosphoryl group to Phosphorylase b, converting it to Phosphorylase a, was named Phosphorylase Kinase. Years later, the first example of a kinase cascade was identified, whereby Protein Kinase A (PKA) phosphorylates Phosphorylase Kinase. At the same time, it was found that PKA inhibited glycogen synthase, which was the first example of a phosphorylation event that resulted in inhibition. In 1969, Lester Reed discovered that pyruvate dehydrogenase was inactivated by phosphorylation, and this discovery was the first clue that phosphorylation might serve as a means of regulation in other metabolic pathways besides glycogen metabolism. In the same year, Tom Langan discovered that PKA phosphorylates histone H1, which suggested phosphorylation might regulate nonenzymatic proteins. The 1970s included the discovery of calmodulin-dependent protein kinases and the finding that proteins can be phosphorylated on more than one amino acid residue. The 1990s may be described as the "decade of protein kinase cascades". During this time, the MAPK/ERK pathway, the JAK kinases (a family of protein tyrosine kinases), and the PIP3-dependent kinase cascade were discovered.[11]
Kinases are classified into broad groups by the substrate they act upon: protein kinases, lipid kinases, carbohydrate kinases. Kinases can be found in a variety of species, from bacteria to mold to worms to mammals.[12] More than five hundred different kinases have been identified in humans.[2] Their diversity and their role in signaling makes them an interesting object of study. Various other kinases act on small molecules such as lipids, carbohydrates, amino acids, and nucleotides, either for signaling or to prime them for metabolic pathways. Specific kinases are often named after their substrates. Protein kinases often have multiple substrates, and proteins can serve as substrates for more than one specific kinase. For this reason protein kinases are named based on what regulates their activity (i.e. Calmodulin-dependent protein kinases). Sometimes they are further subdivided into categories because there are several isoenzymatic forms. For example, type I and type II cyclic-AMP dependent protein kinases have identical catalytic subunits but different regulatory subunits that bind cyclic AMP.[13]
Protein kinases
Protein kinases act on proteins, by phosphorylating them on their serine, threonine, tyrosine, or histidine residues. Phosphorylation can modify the function of a protein in many ways. It can increase or decrease a protein's activity, stabilize it or mark it for destruction, localize it within a specific cellular compartment, and it can initiate or disrupt its interaction with other proteins. The protein kinases make up the majority of all kinases and are widely studied.[14] These kinases, in conjunction with phosphatases, play a major role in protein and enzyme regulation as well as signalling in the cell.
A common point of confusion arises when thinking about the different ways a cell achieves biological regulation. There are countless examples of covalent modifications that cellular proteins can undergo; however, phosphorylation is one of the few reversible covalent modifications. This provided the rationale that phosphorylation of proteins is regulatory. The potential to regulate protein function is enormous given that there are many ways to covalently modify a protein in addition to regulation provided by allosteric control. In his Hopkins Memorial Lecture, Edwin Krebs asserted that allosteric control evolved to respond to signals arising from inside the cell, whereas phosphorylation evolved to respond to signals outside of the cell. This idea is consistent with the fact that phosphorylation of proteins occurs much more frequently in eukaryotic cells in comparison to prokaryotic cells because the more complex cell type evolved to respond to a wider array of signals.[13]
Cyclin dependent kinases
Cyclin dependent kinases (CDKs) are a group of several different kinases involved in regulation of the cell cycle. They phosphorylate other proteins on their serine or threonine residues, but CDKs must first bind to a cyclin protein in order to be active.[15] Different combinations of specific CDKs and cyclins mark different parts of the cell cycle. Additionally, the phosphorylation state of CDKs is also critical to their activity, as they are subject to regulation by other kinases (such as CDK-activating kinase) and phosphatases (such as Cdc25).[16] Once the CDKs are active, they phosphorylate other proteins to change their activity, which leads to events necessary for the next stage of the cell cycle. While they are most known for their function in cell cycle control, CDKs also have roles in transcription, metabolism, and other cellular events.[17]
Because of their key role in the controlling cell division, mutations in CDKs are often found in cancerous cells. These mutations lead to uncontrolled growth of the cells, where they are rapidly going through the whole cell cycle repeatedly.[18] CDK mutations can be found in lymphomas, breast cancer, pancreatic tumors, and lung cancer. Therefore, inhibitors of CDK have been developed as treatments for some types of cancer.[18]
Mitogen-activated protein kinases
MAP kinases (MAPKs) are a family of serine/threonine kinases that respond to a variety of extracellular growth signals. For example, growth hormone, epidermal growth factor, platelet-derived growth factor, and insulin are all considered mitogenic stimuli that can engage the MAPK pathway. Activation of this pathway at the level of the receptor initiates a signaling cascade whereby the Ras GTPase exchanges GDP for GTP. Next, Ras activates Raf kinase (also known as MAPKKK), which activates MEK (MAPKK). MEK activates MAPK (also known as ERK), which can go on to regulate transcription and translation. Whereas RAF and MAPK are both serine/threonine kinases, MAPKK is a tyrosine/threonine kinase.

MAPK can regulate transcription factors directly or indirectly. Its major transcriptional targets include ATF-2, Chop, c-Jun, c-Myc, DPC4, Elk-1, Ets1, Max, MEF2C, NFAT4, Sap1a, STATs, Tal, p53, CREB, and Myc. MAPK can also regulate translation by phosphorylating the S6 kinase in the large ribosomal subunit. It can also phosphorylate components in the upstream portion of the MAPK signalling cascade including Ras, Sos, and the EGF receptor itself.[19]
The carcinogenic potential of the MAPK pathway makes it clinically significant. It is implicated in cell processes that can lead to uncontrolled growth and subsequent tumor formation. Mutations within this pathway alter its regulatory effects on cell differentiation, proliferation, survival, and apoptosis, all of which are implicated in various forms of cancer.[19]
Lipid kinases
Lipid kinases phosphorylate lipids in the cell, both on the plasma membrane as well as on the membranes of the organelles. The addition of phosphate groups can change the reactivity and localization of the lipid and can be used in signal transmission.
Phosphatidylinositol kinases
{{See also|Phosphatidylinositol p
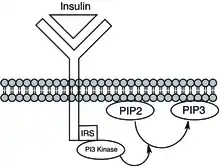
Phosphatidylinositol kinases phosphorylate phosphatidylinositol species, to create species such as phosphatidylinositol 3,4-bisphosphate (PI(3,4)P2), phosphatidylinositol 3,4,5-trisphosphate (PIP3), and phosphatidylinositol 3-phosphate (PI3P). The kinases include phosphoinositide 3-kinase (PI3K), phosphatidylinositol-4-phosphate 3-kinase, and phosphatidylinositol-4,5-bisphosphate 3-kinase. The phosphorylation state of phosphatidylinositol plays a major role in cellular signalling, such as in the insulin signalling pathway, and also has roles in endocytosis, exocytosis and other trafficking events.[20][21] Mutations in these kinases, such as PI3K, can lead to cancer or insulin resistance.[22]
The kinase enzymes increase the rate of the reactions by making the inositol hydroxyl group more nucleophilic, often using the side chain of an amino acid residue to act as a general base and deprotonate the hydroxyl, as seen in the mechanism below.[23] Here, a reaction between adenosine triphosphate (ATP) and phosphatidylinositol is coordinated. The end result is a phosphatidylinositol-3-phosphate as well as adenosine diphosphate (ADP). The enzymes can also help to properly orient the ATP molecule, as well as the inositol group, to make the reaction proceed faster. Metal ions are often coordinated for this purpose.[23]
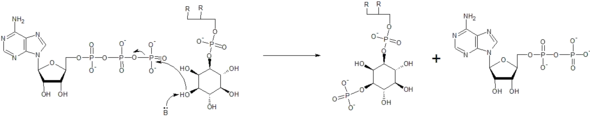
Sphingosine kinases
Sphingosine kinase (SK) is a lipid kinase that catalyzes the conversion of sphingosine to sphingosine-1-phosphate (S1P). Sphingolipids are ubiquitous membrane lipids. Upon activation, sphingosine kinase migrates from the cytosol to the plasma membrane where it transfers a γ phosphate (which is the last or terminal phosphate) from ATP or GTP to sphingosine. The S1P receptor is a GPCR receptor, so S1P has the ability to regulate G protein signaling. The resulting signal can activate intracellular effectors like ERKs, Rho GTPase, Rac GTPase, PLC, and AKT/PI3K. It can also exert its effect on target molecules inside the cell. S1P has been shown to directly inhibit the histone deacetylase activity of HDACs. In contrast, the dephosphorylated sphingosine promotes cell apoptosis, and it is therefore critical to understand the regulation of SKs because of its role in determining cell fate. Past research shows that SKs may sustain cancer cell growth because they promote cellular-proliferation, and SK1 (a specific type of SK) is present at higher concentrations in certain types of cancers.
There are two kinases present in mammalian cells, SK1 and SK2. SK1 is more specific compared to SK2, and their expression patterns differ as well. SK1 is expressed in lung, spleen, and leukocyte cells, whereas SK2 is expressed in kidney and liver cells. The involvement of these two kinases in cell survival, proliferation, differentiation, and inflammation makes them viable candidates for chemotherapeutic therapies.[24]
Carbohydrate kinases
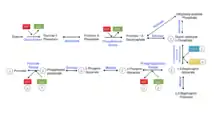
For many mammals, carbohydrates provide a large portion of the daily caloric requirement. To harvest energy from oligosaccharides, they must first be broken down into monosaccharides so they can enter metabolism. Kinases play an important role in almost all metabolic pathways. The figure on the left shows the second phase of glycolysis, which contains two important reactions catalyzed by kinases. The anhydride linkage in 1,3 bisphosphoglycerate is unstable and has a high energy. 1,3-bisphosphogylcerate kinase requires ADP to carry out its reaction yielding 3-phosphoglycerate and ATP. In the final step of glycolysis, pyruvate kinase transfers a phosphoryl group from phosphoenolpyruvate to ADP, generating ATP and pyruvate.
Hexokinase is the most common enzyme that makes use of glucose when it first enters the cell. It converts D-glucose to glucose-6-phosphate by transferring the gamma phosphate of an ATP to the C6 position. This is an important step in glycolysis because it traps glucose inside the cell due to the negative charge. In its dephosphorylated form, glucose can move back and forth across the membrane very easily.[25] Mutations in the hexokinase gene can lead to a hexokinase deficiency which can cause nonspherocytic hemolytic anemia.[26]
Phosphofructokinase, or PFK, catalyzes the conversion of fructose-6-phosphate to fructose-1,6-bisphosphate and is an important point in the regulation of glycolysis. High levels of ATP, H+, and citrate inhibit PFK. If citrate levels are high, it means that glycolysis is functioning at an optimal rate. High levels of AMP stimulate PFK. Tarui's disease, a glycogen storage disease that leads to exercise intolerance, is due to a mutation in the PFK gene that reduces its activity.[27]
Other kinases

Kinases act upon many other molecules besides proteins, lipids, and carbohydrates. There are many that act on nucleotides (DNA and RNA) including those involved in nucleotide interconverstion, such as nucleoside-phosphate kinases and nucleoside-diphosphate kinases.[29] Other small molecules that are substrates of kinases include creatine, phosphoglycerate, riboflavin, dihydroxyacetone, shikimate, and many others.
Riboflavin kinase
Riboflavin kinase catalyzes the phosphorylation of riboflavin to create flavin mononucleotide(FMN). It has an ordered binding mechanism where riboflavin must bind to the kinase before it binds to the ATP molecule.[30] Divalent cations help coordinate the nucleotide.[30] The general mechanism is shown in the figure below.

Riboflavin kinase plays an important role in cells, as FMN is an important cofactor. FMN also is a precursor to flavin adenine dinucleotide(FAD), a redox cofactor used by many enzymes, including many in metabolism. In fact, there are some enzymes that are capable of carrying out both the phosphorylation of riboflavin to FMN, as well as the FMN to FAD reaction.[31] Riboflavin kinase may help prevent stroke, and could possibly be used as a treatment in the future.[32] It is also implicated in infection, when studied in mice.[33]
Thymidine kinase
Thymidine kinase is one of the many nucleoside kinases that are responsible for nucleoside phosphorylation. It phosphorylates thymidine to create thymidine monophosphate (dTMP). This kinase uses an ATP molecule to supply the phosphate to thymidine, as shown below. This transfer of a phosphate from one nucleotide to another by thymidine kinase, as well as other nucleoside and nucleotide kinases, functions to help control the level of each of the different nucleotides.
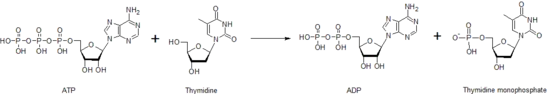
After creation of the dTMP molecule, another kinase, thymidylate kinase, can act upon dTMP to create the diphosphate form, dTDP. Nucleoside diphosphate kinase catalyzes production of thymidine triphosphate, dTTP, which is used in DNA synthesis. Because of this, thymidine kinase activity is closely correlated with the cell cycle and used as a tumor marker in clinical chemistry.[34] Therefore, it can sometime be used to predict patient prognosis.[35] Patients with mutations in the thymidine kinase gene may have a certain type of mitochondrial DNA depletion syndrome, a disease that leads to death in early childhood.[36]
See also
| Wikimedia Commons has media related to Kinases. |
- Activation loop
- Autophosphorylation
- Ca2+/calmodulin-dependent protein kinase
- Cell signaling
- Cyclin-dependent kinase
- G protein-coupled receptor
- Nucleoside-diphosphate kinase
- Phosphatase
- Phosphatidylinositol phosphate kinases
- Phospholipid
- Phosphoprotein
- Phosphorylation
- Phosphotransferase
- Signal transduction
- Thymidine kinase
- Thymidine kinase in clinical chemistry
- Thymidylate kinase
- Wall-associated kinase
References
- Siebold, C; Arnold, I; Garcia-Alles, LF; Baumann, U; Erni, B (Nov 28, 2003). "Crystal structure of the Citrobacter freundii dihydroxyacetone kinase reveals an eight-stranded alpha-helical barrel ATP-binding domain". The Journal of Biological Chemistry. 278 (48): 48236–44. doi:10.1074/jbc.M305942200. PMID 12966101.
- Manning G, Whyte DB, et al. (2002). "The protein kinase complement of the human genome". Science. 298 (5600): 1912–1934. doi:10.1126/science.1075762. PMID 12471243.
- "Kinase". TheFreeDictionary.com
- "History of ATP research milestones from an ATP-related chemistry". Nobelprize.org.
- Reiterer V, Eyers PA, Farhan H (2014). "Day of the dead: pseudokinases and pseudophosphatases in physiology and disease". Trends in Cell Biology. 24 (9): 489–505. doi:10.1016/j.tcb.2014.03.008. PMID 24818526.
- Foulkes DM, Byrne DP and Eyers PA (2017) Pseudokinases: update on their functions and evaluation as new drug targets. Future Med Chem. 9(2):245-265
- Samarasinghe, Buddhini. "Hallmarks of Cancer 1". Scientific American.
- Bleeker, FE; Lamba, S; Zanon, C; Molenaar, RJ; Hulsebos, TJ; Troost, D; van Tilborg, AA; Vandertop, WP; Leenstra, S; van Noorden, CJ; Bardelli, A (26 September 2014). "Mutational profiling of kinases in glioblastoma". BMC Cancer. 14: 718. doi:10.1186/1471-2407-14-718. PMC 4192443. PMID 25256166.
- Lahiry, Piya; Torkamani, Ali; Schork, Nicholas J.; Hegele, Robert A. (January 2010). "Kinase mutations in human disease: interpreting genotype–phenotype relationships". Nature Reviews Genetics. 11 (1): 60–74. doi:10.1038/nrg2707. PMID 20019687.
- Krebs, EG (Jul 5, 1983). "Historical perspectives on protein phosphorylation and a classification system for protein kinases". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 302 (1108): 3–11. doi:10.1098/rstb.1983.0033. PMID 6137005.
- Corbellino, M; Poirel, L; Aubin, JT; Paulli, M; Magrini, U; Bestetti, G; Galli, M; Parravicini, C (Jun 1996). "The role of human herpesvirus 8 and Epstein-Barr virus in the pathogenesis of giant lymph node hyperplasia (Castleman's disease)". Clinical Infectious Diseases. 22 (6): 1120–1. doi:10.1093/clinids/22.6.1120. PMID 8783733.
- Scheeff, Eric D.; Bourne, Philip E. (2005). "Structural Evolution of the Protein Kinase–Like Superfamily". PLoS Computational Biology. 1 (5): e49. doi:10.1371/journal.pcbi.0010049. PMC 1261164. PMID 16244704.
- Krebs, EG; Tan, ST; Carrow, DJ; Watts, MK (Oct 1985). "The phosphorylation of proteins: a major mechanism for biological regulation. Fourteenth Sir Frederick Gowland Hopkins memorial lecture". Biochemical Society Transactions. 13 (5): 813–20. doi:10.1042/bst0130813. PMID 2998902.
- Manning, G; Whyte, DB; Martinez, R; Hunter, T; Sudarsanam, S (Dec 6, 2002). "The protein kinase complement of the human genome". Science. 298 (5600): 1912–34. doi:10.1126/science.1075762. PMID 12471243.
- Harper, J. W.; Adams, P. D. (August 2001). "Cyclin-Dependent Kinases". Chemical Reviews. 101 (8): 2511–2526. doi:10.1021/cr0001030. PMID 11749386.
- Karp, Gerald (2010). Cell and molecular biology : concepts and experiments (6th ed.). Hoboken, NJ: John Wiley. ISBN 9780470483374.
- Lim, S.; Kaldis, P. (16 July 2013). "Cdks, cyclins and CKIs: roles beyond cell cycle regulation". Development. 140 (15): 3079–3093. doi:10.1242/dev.091744. PMID 23861057.
- Canavese, Miriam; Santo, Loredana; Raje, Noopur (1 May 2012). "Cyclin dependent kinases in cancer: Potential for therapeutic intervention". Cancer Biology & Therapy. 13 (7): 451–457. doi:10.4161/cbt.19589. PMID 22361734.
- Garrington, TP; Johnson, GL (Apr 1999). "Organization and regulation of mitogen-activated protein kinase signaling pathways". Current Opinion in Cell Biology. 11 (2): 211–8. doi:10.1016/s0955-0674(99)80028-3. PMID 10209154.
- Sun, Yue; Thapa, Narendra; Hedman, Andrew C.; Anderson, Richard A. (June 2013). "Phosphatidylinositol 4,5-bisphosphate: Targeted production and signaling". BioEssays. 35 (6): 513–522. doi:10.1002/bies.201200171. PMC 3882169. PMID 23575577.
- Heath, CM; et al. (2003). "Lipid Kinases Play Crucial and Multiple Roles in Membrane Trafficking and Signalling" (PDF). Histology and Histopathology. 18: 989–998.
- Cantley, Lewis C (2012). "PI 3-kinase and disease". BMC Proceedings. 6 (Suppl 3): O2. doi:10.1186/1753-6561-6-S3-O2.
- Miller, S.; Tavshanjian, B.; Oleksy, A.; Perisic, O.; Houseman, B. T.; Shokat, K. M.; Williams, R. L. (25 March 2010). "Shaping Development of Autophagy Inhibitors with the Structure of the Lipid Kinase Vps34". Science. 327 (5973): 1638–1642. doi:10.1126/science.1184429. PMC 2860105. PMID 20339072.
- Neubauer, Heidi A.; Pitson, Stuart M. (November 2013). "Roles, regulation and inhibitors of sphingosine kinase 2". FEBS Journal. 280 (21): 5317–5336. doi:10.1111/febs.12314. PMID 23638983.
- Holzer, H; Duntze, W (1971). "Metabolic regulation by chemical modification of enzymes". Annual Review of Biochemistry. 40: 345–74. doi:10.1146/annurev.bi.40.070171.002021. PMID 4399446.
- "Nonspherocytic hemolytic anemia due to hexokinase deficiency".
- "Phosphofructokinase Deficiency Glycogen Storage Disease".
- Bauer, S; Kemter, K; Bacher, A; Huber, R; Fischer, M; Steinbacher, S (Mar 7, 2003). "Crystal structure of Schizosaccharomyces pombe riboflavin kinase reveals a novel ATP and riboflavin-binding fold". Journal of Molecular Biology. 326 (5): 1463–73. doi:10.1016/s0022-2836(03)00059-7. PMID 12595258.
- Pratt, Donald Voet, Judith G. Voet, Charlotte W. (2008). Fundamentals of biochemistry : life at the molecular level (3rd ed.). Hoboken, NJ: Wiley. ISBN 9780470129302.
- Karthikeyan, S; Zhou, Q; Osterman, AL; Zhang, H (Nov 4, 2003). "Ligand binding-induced conformational changes in riboflavin kinase: structural basis for the ordered mechanism". Biochemistry. 42 (43): 12532–8. doi:10.1021/bi035450t. PMID 14580199.
- Galluccio, M; Brizio, C; Torchetti, EM; Ferranti, P; Gianazza, E; Indiveri, C; Barile, M (Mar 2007). "Over-expression in Escherichia coli, purification and characterization of isoform 2 of human FAD synthetase". Protein Expression and Purification. 52 (1): 175–81. doi:10.1016/j.pep.2006.09.002. PMID 17049878.
- Zou, YX; Zhang, XH; Su, FY; Liu, X (Oct 2012). "Importance of riboflavin kinase in the pathogenesis of stroke". CNS Neuroscience & Therapeutics. 18 (10): 834–40. doi:10.1111/j.1755-5949.2012.00379.x. PMC 6493343. PMID 22925047.
- Brijlal, Sangeetha; Lakshmi, A. V; Bamji, Mahtab S.; Suresh, P. (9 March 2007). "Flavin metabolism during respiratory infection in mice". British Journal of Nutrition. 76 (3): 453–62. doi:10.1079/BJN19960050. PMID 8881717.
- Aufderklamm, S; Todenhöfer, T; Gakis, G; Kruck, S; Hennenlotter, J; Stenzl, A; Schwentner, C (Mar 2012). "Thymidine kinase and cancer monitoring". Cancer Letters. 316 (1): 6–10. doi:10.1016/j.canlet.2011.10.025. PMID 22068047.
- Topolcan, Ondrej; Holubec, Lubos (February 2008). "The role of thymidine kinase in cancer diseases". Expert Opinion on Medical Diagnostics. 2 (2): 129–141. doi:10.1517/17530059.2.2.129. PMID 23485133.
- Gotz, A.; Isohanni, P.; Pihko, H.; Paetau, A.; Herva, R.; Saarenpaa-Heikkila, O.; Valanne, L.; Marjavaara, S.; Suomalainen, A. (21 June 2008). "Thymidine kinase 2 defects can cause multi-tissue mtDNA depletion syndrome". Brain. 131 (11): 2841–2850. doi:10.1093/brain/awn236. PMID 18819985.
