Abexinostat
Abexinostat (INN,[1] formerly PCI-24781) is an experimental drug candidate for cancer treatment.[2] It was developed by Pharmacyclics and licensed to Xynomic. and is in Phase II clinical trials for B-cell lymphoma.[3] Pre-clinical study suggests the potential for treatment of different types of cancer as well.[4][5][6][7]
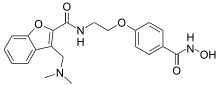 | |
| Names | |
|---|---|
| IUPAC name
3-[(Dimethylamino)methyl]-N-{2-[4-(hydroxycarbamoyl)phenoxy]ethyl}-1-benzofuran-2-carboxamide | |
| Other names
PCI-24781; CRA-024781 | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.241.399 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C21H23N3O5 | |
| Molar mass | 397.431 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Abexinostat exerts its effect as a pan-histone deacetylase inhibitor[8][9] and inhibits RAD51, which is involved in repairing DNA double strand breaks.[10]
References
- WHO Drug Information, Vol. 25, No. 2, 2011
- Abexinostat, NCI Cancer Dictionary
- Abexinostat HCl (PCI-24781), PanHDAC-inhibitor Archived 2013-10-27 at the Wayback Machine, Pharmacyclics
- Bhalla, S; Balasubramanian, S; David, K; Sirisawad, M; Buggy, J; Mauro, L; Prachand, S; Miller, R; Gordon, LI; Evens, AM (2009). "PCI-24781 induces caspase and reactive oxygen species-dependent apoptosis through NF-kappaB mechanisms and is synergistic with bortezomib in lymphoma cells". Clinical Cancer Research. 15 (10): 3354–65. doi:10.1158/1078-0432.CCR-08-2365. PMC 2704489. PMID 19417023.
- Lopez, G; Liu, J; Ren, W; Wei, W; Wang, S; Lahat, G; Zhu, QS; Bornmann, WG; McConkey, DJ; Pollock, RE; Lev, DC (2009). "Combining PCI-24781, a novel histone deacetylase inhibitor, with chemotherapy for the treatment of soft tissue sarcoma". Clinical Cancer Research. 15 (10): 3472–83. doi:10.1158/1078-0432.CCR-08-2714. PMID 19417021.
- Rivera-Del Valle, N; Gao, S; Miller, CP; Fulbright, J; Gonzales, C; Sirisawad, M; Steggerda, S; Wheler, J; Balasubramanian, S; Chandra, J (2010). "PCI-24781, a Novel Hydroxamic Acid HDAC Inhibitor, Exerts Cytotoxicity and Histone Alterations via Caspase-8 and FADD in Leukemia Cells". International Journal of Cell Biology. 2010: 207420. doi:10.1155/2010/207420. PMC 2817379. PMID 20145726.
- Yang, C; Choy, E; Hornicek, FJ; Wood, KB; Schwab, JH; Liu, X; Mankin, H; Duan, Z (2011). "Histone deacetylase inhibitor (HDACI) PCI-24781 potentiates cytotoxic effects of doxorubicin in bone sarcoma cells". Cancer Chemotherapy and Pharmacology. 67 (2): 439–46. doi:10.1007/s00280-010-1344-7. PMID 20461381.
- Buggy, JJ; Cao, ZA; Bass, KE; Verner, E; Balasubramanian, S; Liu, L; Schultz, BE; Young, PR; Dalrymple, SA (2006). "CRA-024781: A novel synthetic inhibitor of histone deacetylase enzymes with antitumor activity in vitro and in vivo". Molecular Cancer Therapeutics. 5 (5): 1309–17. doi:10.1158/1535-7163.MCT-05-0442. PMID 16731764.
- Adimoolam, S; Sirisawad, M; Chen, J; Thiemann, P; Ford, JM; Buggy, JJ (2007). "HDAC inhibitor PCI-24781 decreases RAD51 expression and inhibits homologous recombination". Proceedings of the National Academy of Sciences of the United States of America. 104 (49): 19482–7. doi:10.1073/pnas.0707828104. PMC 2148315. PMID 18042714.
- "NCI Drug Dictionary". National Cancer Institute.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.