Setmelanotide
Setmelanotide, sold under the brand name Imcivree, is a medication for the treatment of obesity.[1][2]
 | |
| Clinical data | |
|---|---|
| Trade names | Imcivree |
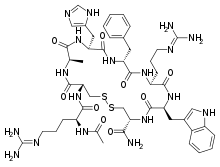
| Other names | RM-493; BIM-22493; IRC-022493; N2-Acetyl-L-arginyl-L-cysteinyl-D-alanyl-L-histidyl-D-phenylalanyl-L-arginyl-L-tryptophyl-L-cysteinamide, cyclic (2-8)-disulfide |
| License data | |
| Routes of administration | Subcutaneous |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C49H68N18O9S2 |
| Molar mass | 1117.32 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
The most common side effects include injection site reactions, skin hyperpigmentation (skin patches that are darker than surrounding skin), headache and gastrointestinal side effects (such as nausea, diarrhea, and abdominal pain), among others.[2] Spontaneous penile erections in males and adverse sexual reactions in females have occurred with treatment.[2] Depression and suicidal ideation have also occurred with setmelanotide.[2]
Setmelanotide was approved for medical use in the United States in November 2020.[3] The U.S. Food and Drug Administration (FDA) considers it to be a first-in-class medication.[4]
Medical uses
Setmelanotide is indicated for chronic weight management (weight loss and weight maintenance for at least one year) in people six years and older with obesity due to three rare genetic conditions: pro-opiomelanocortin (POMC) deficiency, proprotein subtilisin/kexin type 1 (PCSK1) deficiency, and leptin receptor (LEPR) deficiency confirmed by genetic testing demonstrating variants in POMC, PCSK1, or LEPR genes considered pathogenic (causing disease), likely pathogenic, or of uncertain significance.[2] Setmelanotide is the first FDA-approved treatment for these genetic conditions.[2]
Setmelanotide is not approved for obesity due to suspected POMC, PCSK1, or LEPR deficiency with variants classified as benign (not causing disease) or likely benign or other types of obesity, including obesity associated with other genetic syndromes and general (polygenic) obesity.[2]
Setmelanotide binds to and activates MC4 receptors in the paraventricular nucleus (PVN) of the hypothalamus and in the lateral hypothalamic area (LHA), areas involved in the regulation of appetite, and this action is thought to underlie its appetite suppressant effects.[5] In addition to reducing appetite, setmelanotide increases resting energy expenditure in both obese animals and humans.[6] Importantly, unlike certain other MC4 receptor agonists, such as LY-2112688, setmelanotide has not been found to produce increases in heart rate or blood pressure.[7]
Setmelanotide has been reported to possess the following activity profile (cAMP, EC50): MC4 (0.27 nM) > MC3 (5.3 nM) ≈ MC1 (5.8 nM) > MC5 (1600 nM) ≟ MC2 (>1000 nM).[8] (19.6-fold selectivity for MC4 over MC3, the second target of highest activity.)
History
Setmelanotide was evaluated in two one-year studies.[2] The first study enrolled participants with obesity and confirmed or suspected POMC or PCSK1 deficiency while the second study enrolled participants with obesity and confirmed or suspected LEPR deficiency; all participants were six years or older.[2] The effectiveness of setmelanotide was determined by the number of participants who lost more than ten percent of their body weight after a year of treatment.[2]
The effectiveness of setmelanotide was assessed in 21 participants, ten in the first study and eleven in the second.[2] In the first study, 80 percent of participants with POMC or PCSK1 deficiency lost ten percent or more of their body weight.[2] In the second study, 46 percent of participants with LEPR deficiency lost ten percent or more of their body weight.[2]
The study also assessed the maximal (greatest) hunger in sixteen participants over the previous 24 hours using an eleven-point scale in participants twelve years and older.[2] In both studies, some, but not all, of participants' weekly average maximal hunger scores decreased substantially from their scores at the beginning of the study.[2] The degree of change was highly variable among participants.[2]
The U.S. Food and Drug Administration (FDA) granted the application for setmelanotide orphan disease designation, breakthrough therapy designation, and priority review.[2] The FDA granted the approval of Imcivree to Rhythm Pharmaceutical, Inc.[2][3]
Research
Setmelanotide is a peptide drug and investigational anti-obesity medication which acts as a selective agonist of the MC4 receptor.[9][7] Its peptide sequence is Ac-Arg-Cys(1)-D-Ala-His-D-Phe-Arg-Trp-Cys(1)-NH2. It was first discovered at Ipsen and is being developed by Rhythm Pharmaceuticals for the treatment of obesity and diabetes.[9] In addition, Rhythm Pharmaceuticals is conducting trials of setmelanotide for the treatment of Prader–Willi syndrome (PWS), a genetic disorder which includes MC4 receptor deficiency and associated symptoms such as excessive appetite and obesity.[10] As of December 2014, the drug is in phase II clinical trials for obesity and PWS.[9][11][12] So far, preliminary data has shown no benefit of Setmelanotide in Prader-Willi syndrome.[13]
References
- "Imcivree- setmelanotide solution". DailyMed. Retrieved 25 December 2020.
- "FDA approves first treatment for weight management for people with certain rare genetic conditions". U.S. Food and Drug Administration (FDA) (Press release). 27 November 2020. Retrieved 27 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - "Drug Approval Package: Imcivree". U.S. Food and Drug Administration (FDA). 23 December 2020. Retrieved 18 January 2021.
- "New Drug Therapy Approvals 2020". U.S. Food and Drug Administration (FDA). 31 December 2020. Retrieved 17 January 2021.
- Kim GW, Lin JE, Blomain ES, Waldman SA (January 2014). "Antiobesity pharmacotherapy: new drugs and emerging targets". Clinical Pharmacology and Therapeutics. 95 (1): 53–66. doi:10.1038/clpt.2013.204. PMC 4054704. PMID 24105257.
- Chen KY, Muniyappa R, Abel BS, Mullins KP, Staker P, Brychta RJ, et al. (April 2015). "RM-493, a melanocortin-4 receptor (MC4R) agonist, increases resting energy expenditure in obese individuals". The Journal of Clinical Endocrinology and Metabolism. 100 (4): 1639–45. doi:10.1210/jc.2014-4024. PMC 4399297. PMID 25675384.
- Kievit P, Halem H, Marks DL, Dong JZ, Glavas MM, Sinnayah P, et al. (February 2013). "Chronic treatment with a melanocortin-4 receptor agonist causes weight loss, reduces insulin resistance, and improves cardiovascular function in diet-induced obese rhesus macaques". Diabetes. 62 (2): 490–7. doi:10.2337/db12-0598. PMC 3554387. PMID 23048186.
- Muniyappa R, Chen K, Brychta R, Abel B, Mullins K, Staker P, et al. (June 2014). "A Randomized, Double-Blind, Placebo-Controlled, Crossover Study to Evaluate the Effect of a Melanocortin Receptor 4 (MC4R) Agonist, RM-493, on Resting Energy Expenditure (REE) in Obese Subjects" (PDF). Endocrine Reviews. Rhythm Pharmaceuticals. 35 (3). Retrieved 2015-05-21.
- Lee EC, Carpino PA (2015). "Melanocortin-4 receptor modulators for the treatment of obesity: a patent analysis (2008-2014)". Pharmaceutical Patent Analyst. 4 (2): 95–107. doi:10.4155/ppa.15.1. PMID 25853469.
- "Obesity and Diabetes Caused by Genetic Deficiencies in the MC4 Pathway". Rhythm Pharmaceuticals. Retrieved 2015-05-21.
- Jackson VM, Price DA, Carpino PA (August 2014). "Investigational drugs in Phase II clinical trials for the treatment of obesity: implications for future development of novel therapies". Expert Opinion on Investigational Drugs. 23 (8): 1055–66. doi:10.1517/13543784.2014.918952. PMID 25000213. S2CID 23198484.
- "RM-493: A First-in-Class, Phase 2-Ready MC4 Agonist: A New Drug Class for the Treatment of Obesity and Diabetes". Rhythm Pharmaceuticals. Archived from the original on 2015-06-14. Retrieved 2015-05-21.
- Duis J, van Wattum PJ, Scheimann A, Salehi P, Brokamp E, Fairbrother L, et al. (March 2019). "A multidisciplinary approach to the clinical management of Prader-Willi syndrome". Molecular Genetics & Genomic Medicine. 7 (3): e514. doi:10.1002/mgg3.514. PMC 6418440. PMID 30697974.
External links
- "Setmelanotide". Drug Information Portal. U.S. National Library of Medicine.