Neuromyelitis optica spectrum disorder
Neuromyelitis optica spectrum disorders (NMOSD) is an etiologically heterogeneous syndrome predominantly characterized by acute inflammation of the optic nerve (optic neuritis, ON) and the spinal cord (myelitis).[1][2] Episodes of ON and myelitis can be simultaneous or successive. A relapsing disease course is common, especially in untreated patients.[1][3] In more than 80% of cases, NMO is caused by immunoglobulin G autoantibodies to aquaporin 4 (anti-AQP4), the most abundant water channel protein in the central nervous system. A subset of anti-AQP4-negative cases is associated with antibodies to myelin oligodendrocyte glycoprotein (anti-MOG).[1][3] Rarely, NMO may occur in the context of other autoimmune diseases (e.g. connective tissue disorders, paraneoplastic syndromes) or infectious diseases. In some cases, the etiology remains unknown (idiopathic NMO).
| Neuromyelitis optica spectrum disorders | |
|---|---|
| Other names | Neuromyelitis optica (NMO), Devic's disease, Devic's syndrome |
| Specialty | Neurology, ophthalmology |
| Symptoms | Vision loss, sensory loss, weakness, bladder dysfunction |
| Usual onset | Median: age 40 for AQP4-IgG, age 31 for MOG-IgG[1] |
| Types | AQP4-IgG-positive, MOG-IgG-positive (recurrent, monophasic)[1] |
| Risk factors | Female sex, genetic factors[1] |
| Diagnostic method | Symptoms, blood antibody titers, MRI |
| Differential diagnosis | Multiple sclerosis, various autoimmune disorders |
| Medication | Eculizumab, inebilizumab, satralizumab, rituximab, methylprednisolone, azathioprine, cellCept, mitoxantrone, methotrexate, intravenous immunoglobulin, cyclophosphamide |
| Frequency | Up to 1/10,000[1] |
Multiple sclerosis (MS) and NMO can be similar in clinical and radiological presentation, and MS may very rarely present with an NMO-like phenotype (e.g. in patients with long-standing MS resulting in confluent spinal cord lesions mimicking the long spinal cord lesions typically seen in MS). In consequence, NMO was wrongly considered a clinical variant of MS in the past. However, NMO is not caused by MS in the vast majority of cases but differs from MS substantially in terms of pathogenesis, clinical presentation, magnetic resonance imaging, cerebrospinal fluid findings, disease course and prognosis.[1]
Signs and symptoms
The signs and symptoms of NMOSD are dependent on which neurologic structures the disease affects, and, to some extent, which antibodies are involved. Signs and symptoms usually follow a relapsing and remitting course, but occasional can be progressive (monophasic). Deficits can be temporary or permanent, the latter especially in the absence of treatment.
The most common initial manifestation of the disease is inflammation of the spinal cord (myelitis).[3] Myelitis causes spinal cord dysfunction, which can cause muscle weakness, reduced sensation, or loss of bladder and bowel control as well as erectile dysfunction.[1][3] Not seldom, the myelitis is transverse, meaning it affects an entire cross-section of the spinal cord, causing bilateral symptoms.
The second most common initial manifestation of the disease is inflammation of the optic nerve and/or optic chiasm (optic neuritis, ON).[3] ON may lead to varying degrees as visual impairment with decreased visual acuity, although visual field defects, or loss of color vision may occur in isolation or prior to formal loss of visual acuity. Compared to idiopathic ON and ON due to multiple sclerosis (MS), ON due to NMOSD more often has severe visual loss at onset, bilateral involvement, and permanency of visual deficits.[3]
Classically, NMO included only symptoms of myelitis and ON.[3] However, with the discovery of disease-causing antibodies, a broader spectrum of disease manifestations have been grouped with NMO into the diagnosis of NMOSD.[3] Less commonly than the spinal cord and optic nerve, NMOSD can affect the brainstem.[3] Lesions in the brainstem or upper cervical spinal cord can cause respiratory insufficiency. Lesions in area postrema of the medulla oblongata can cause vomiting or hiccups, as well as and pain and tonic spasms.[1][3] Additional brain lesions are common but often asymptomatic (though cognitive deficits as well as depression may be underdiagnosed sequalae). Lesions may also affect the diencephalon (mostly in AQP4-IgG NMOSD).[1][3]
Causes
NMOSD is caused by an autoimmune attack on the nervous system. In more than 80% of cases, IgG autoantibodies against aquaporin-4 (anti-AQP4+) are the cause, and in 10-40% of the remaining cases, IgG antibodies against MOG are the cause.[1] The cause of remaining cases is still unknown, and it is likely heterogeneous.[4][5]
Why autoimmunity develops is largely unknown. Multiple genetic and environmental factors are known to increase risk of developing NMOSD. The strongest risk factor is female sex, especially in AQP4-IgG-positive NMOSD.[1] Multiple human leukocyte antigen (HLA) alleles are associated with NMOSD.[1]
NMO was associated in the past with many systemic diseases. Some researchers have pointed out that some other cases could be paraneoplastic.[6] It seems also clear that lupus can produce NMO-IgG autoantibodies, leading to some cases of lupus-derived NMO.[7]
The discovery of NMO-IgG (anti-AQP4) has opened a new way into the research for the causes.
Pathophysiology

Anti-AQP4+ variants
NMOSD is usually caused by autoantibodies targeting aquaporin 4 (AQP4), a channel protein in the cell membrane that allows water to cross.[8] AQP4 monomers form tetramers, and the tetramers aggregate.[3] AQP4 is found in astrocytes, which are the basis for the glymphatic system.[9] Thus, NMOSD involving AQP4-IgG can be considered an astrocytopathy[10] or autoimmune astrocytic channelopathy, since the astrocytes are semi-selectively destroyed.[11]
The astrocytes surround the blood–brain barrier (BBB), a system responsible for preventing substances in the blood from entering the brain. For antibodies from the blood to reach astrocytes in the CNS, they must cross the BBB, the mechanism of which is not completely known. Some reports point to the metalloproteinase-2 and interleukin-6 as culprits for the BBB failure.[12] There is broad consensus that AQP4/NMO-IgG initially enters the brain via BBB-deficient sites such as area postrema, where access to CSF occurs.[13] In any case, anti-AQP4 is produced mainly intrathecally.[14]
Within astrocytes, AQP4 is primarily found in astrocytic foot processes that abut blood vessels and the lining of the brain (meninges).[1] NMOSD brain lesions, as seen under a microscopic, show IgG, IgM, inflammatory cells, and complement deposits around blood vessels.[1] AQP4-IgG is a member of the IgG1 immunoglobulin family, which is an activator of the complement system, which seems to play an integral part in the autoimmune response.[1] There is a loss of astrocytes, and sometimes also a loss of neurons and oligodendrocytes. Loss of cells other than astrocytes is a consequence of collateral inflammatory damage or astroctye dysfunction.[1]
NMOSD selectively affects the optic nerve, spinal cord, and brain stem. This selectivity can be explained by the increased amount of AQP4 in these structures, and furthermore by the increased amount of AQP4 aggregates in the optic nerve and spinal cord.[1] The increased BBB permeability at the area postrema helps explain involvement there.[1] AQP4 is present in tissues outside of the central nervous system (eg: kidneys), but these tissues aren't affected in NMOSD, at least in part because of the presence of autoimmune downregulators outside of the central nervous system.[1]
In NMOSD, areas of brain tissue that appear normal in conventional MRI can show damage in diffusion tensor imaging, although less so compared to multiple sclerosis (MS).[15]
Most research into the pathology of NMO has focused on the spinal cord. The damage can range from inflammatory demyelination to necrotic damage of the white and grey matters. The inflammatory lesions in NMO have been classified as type II lesions (complement-mediated demyelination), but they differ from MS pattern II lesions in their prominent perivascular distribution. Therefore, the pattern of inflammation is often quite distinct from that seen in MS.[8][16]
AQP4-IgG levels are coarsely correlated with NMOSD disease activity, generally increasing before relapse and declining during remission, with higher levels being correlated to more severe disease manifestation.[1]
NMO-IgG-negative cases are less understood. It seems that astrocytes are spared in these IgG negative cases.[17]
Anti-MOG+ variants
The second most frequent autoantibody in NMO is MOG-IgG, which targets myelin oligodendrocyte glycoprotein (MOG). MOG is an integral membrane glycoprotein found on the surface of oligodendrocytes and the outermost surface of myelin sheaths.[1] Its function is not entirely known.[1] MOG-IgG is produced outside the central nervous system (CNS) despite MOG existing only in the CNS (with the BBB separating the two), leading to the hypothesis that MOG drained via cerebral spinal fluid into lymph nodes causes autoimmune reaction formation.[1]
MOG-IgG-positive NMOSD brain lesions, as seen under a microscopic, show demyelination with preservation of oligodendrocytes and axons, presence of inflammatory cells, and IgG and complement deposits.[1]
MOG-IgG levels coarsely correlate with disease severity, with levels being higher during active disease, and higher levels being associated with more severe disease manifestation.[1]
Antibodies against MOG are considered mostly absent in similar diseases like MS.[18] Therefore, it can be said that anti-MOG is a group contained inside AQP4-IgG-negative NMOSD.[19]
Together with the anti-AQP4 they form the wider part of the NMO spectrum. They classify the NMO cases in four classes, according to the presence or absence of any of these two main auto-antibodies.[20]
The clinical course and the response to therapy is different for these groups, showing a better prognosis for those in the NMO-Ab(−)/MOG-Ab(−) group, and a worse prognosis for those in the NMO-Ab(+)/MOG-Ab(+) group.[20] The MOG-related NMO can be radiologically identified by the conus involvement. Myelin-oligodendrocyte glycoprotein antibody–positive patients were more likely to have conus involvement on spinal magnetic resonance imaging.[21]
Diagnosis
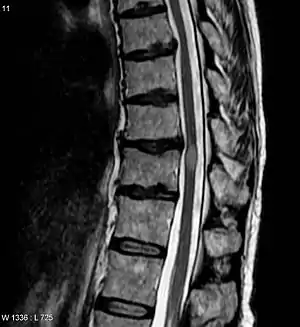
NMOSD is diagnosed using consensus clinical criteria, which have undergone multiple revisions, most recently in 2015.[22][23]
Diagnostic criteria are more relaxed for seropositive AQP4-IgG cases than they are for seronegative AQP4-IgG cases. If AQP4-IgG is detected, then 1 core clinical criterion, along with the ruling out of alternative diagnoses, is sufficient for NMOSD diagnosis.[24]
If AQP4-IgG is undetected or its status unknown, 2 core clinical criteria, each with supportive MRI findings, along with the ruling out of alternative diagnoses, are needed for NMOSD diagnosis.[25]
| Core criteria | Additional MRI findings for absent/unknown AQP4-IgG |
|---|---|
| Optic neuritis | Either 1) brain MRI showing normal findings or only nonspecific white matter lesions, or 2) optic nerve MRI showing T2-hyperintesity, or T1 enhancing lesion, greater than 1/2 optic nerve length or involving optic chiasm |
| Acute myelitis | intramedullary lesion > 3 contiguous segments, or spinal atrophy ≥ 3 contiguous segments |
| Area Postrema Syndrome (prolonged episodes of hiccuping or vomiting/nausea) | dorsal medulla/area postrema lesions |
| Acute brainstem syndrome | periependymal brainstem lesions |
| Symptomatic narcolepsy/acute diencephalic clinical syndrome with a MRI showing diencephalon lesion(s) | None additional |
| Symptomatic cerebral syndrome with NSMOD-typical brain lesion(s) | None additional |
Rarely, it has been reported that some courses of anti-NMDAR are consistent with NMO.[26] Preliminary reports suggest that other autoantibodies may play a role in rare cases of NMO.[27][28]
NMOSD with MOG-IgG is considered a manifestation of anti-MOG associated encephalomyelitis.[29]
Spectrum constituents
After the development of the NMO-IgG test, the spectrum of disorders comprising NMO was expanded. The spectrum is now believed to consist of:
- Standard NMO, according to the diagnostic criteria described above
- Limited forms of NMO, such as single or recurrent events of longitudinally extensive myelitis, and bilateral simultaneous or recurrent optic neuritis
- Asian optic-spinal multiple sclerosis (OSMS), or AQP4+ OSMS. This variant can present brain lesions like MS,[30] but it should not be confused with an AQP4-negative form of inflammatory demyelinating diseases of the central nervous system spectrum, sometimes called optic-spinal MS.
- Longitudinally extensive myelitis or optic neuritis associated with systemic autoimmune disease
- Optic neuritis or myelitis associated with lesions in specific brain areas such as the hypothalamus, periventricular nucleus, and brainstem[31]
- NMO-IgG negative NMO: AQP4 antibody-seronegative NMO poses a diagnostic challenge.[32][33] Some cases could be related to anti-myelin oligodendrocyte glycoprotein (MOG) autoantibodies.[19]
Differential diagnosis
AQP4-Ab-negative NMO presents problems for diagnosis. The behavior of the oligoclonal bands can help to establish a more accurate diagnosis. Oligoclonal bands in NMO are rare and they tend to disappear after the attacks, while in MS they are nearly always present and persistent.[34] It is important to notice for differential diagnosis that, though uncommon, it is possible to have longitudinal lesions in MS.[35]
Other problem for diagnosis is that AQP4-ab in MOG-ab levels can be too low to be detected. Some additional biomarkers have been proposed.[36][37]
NMO differs from MS in that it usually has more severe sequelae after an acute episode than standard MS, MS infrequently presents as transverse myelitis, and oligoclonal bands in the CSF, as well as white matter lesions on brain MRI, are uncommon in NMO, but occur in over 90% of MS patients.[38]
Recently, AQP4 has been found to distinguish standard MS from NMO, but as MS is a heterogeneous condition,[39] and some MS cases are reported to be Kir4.1 channelopathies[40] (autoimmunity against the potassium channels) it is still possible to consider NMO as part of the MS spectrum. Besides, some NMO-AQP4(−) variants are not astrocytopathic, but demyelinating.[41]
Tumefactive demyelinating lesions in NMO are not usual, but they have been reported to appear in several cases mistakenly treated with interferon beta.[42]
Also an overlap with the Sjögren syndrome has been reported.[43]
Evolution of diagnostic criteria
Since the discovery of AQP4 autoantibody, it has been found that it appears also in patients with NMO-like symptoms that do not fulfill the clinical requirements to be diagnosed NMO (recurrent and simultaneous optic nerve and spinal cord inflammation).[44]
The term NMOSD (neuromyelitis optica spectrum disorders) has been designed to allow incorporation of cases associated with non-AQP4 biomarkers.[23] Therefore, it includes all the clinical variants due to anti-AQP4 plus other non-related but clinically similar syndromes like anti-MOG associated encephalomyelitis. Some cases with MOG+ and AQP4+ antibodies have been found.[23]
The collection of these condition has been named "neuromyelitis optica spectrum disorders" (NMOSD) and they are expected to respond to the same treatments as standard NMO.[45] Some authors propose to use the name "autoimmune aquaporin-4 channelopathy" for these diseases,[5] while others prefer a more generic term "AQP4-astrocytopathy" that includes also problems in AQP4 with a non-autoimmune origin.[46]
Treatment

There is no cure for NMO, but it is treatable. Some patients recover, but many are left with impairment of vision and limbs, which can be severe in some cases.[47]
Attacks
Long term neurologic deficits are the cumulative effects of acute attacks, empathizing the importance of acute treatment.[1] Traditionally, attacks have been treated with short courses (3–5 days) of high dosage intravenous corticosteroids, like methylprednisolone IV (Solu-Medrol).[48] Early initiation of treatment with steroids have shown to improve vision-related outcomes after acute attacks.[1][49] However, there is still is no high level evidence for steroids affecting long-term outcomes; this treatment strategy was borrowed from similar diseases (idiopathic optic neuritis and multiple sclerosis).[49][48]
Plasmapheresis can be an effective treatment[31] when attacks progress after the administration of corticosteroids or do not respond to corticosteroids. This treatment involves your own blood being pumped out of your body, your own blood cells being removed from the plasma and being mixed with a solution, then the new blood mixture being pumped back into your body.[47]
Secondary prevention
Treatment, in order to prevent relapses for NMO, is generally employed but the exact duration of prophylaxis is still debated.[50]
FDA-approved pharmaceuticals
FDA-approved pharmaceuticals for AQP4-IgG-positive NMOSD, shown to be effective in phase III clinical trials, became first available in 2019.[1] As of 2020, they are among the most expensive drugs worldwide.[1] They are not available in pill form, which, with their price, reduces their accessibility.[1] These new drugs' effectiveness in AQP4-IgG-negative NMOSD is unknown.[1]
| Drug (brand) | Brand | Date of FDA approval | Mechanism of action | Note |
|---|---|---|---|---|
| Eculizumab | Soliris | 2019 | Monoclonal antibody against complement protein C5 | Approved for AQP4-IgG-positive NMOSD[51] |
| Inebilizumab | Uplizna | 2020 June | Monoclonal antibody against CD19+ B cells | [52] |
| Satralizumab | Enspryng | 2020 August | Monoclonal antibody against IL-6 | Approved forAQP4-IgG-positive NMOSD[53][54] |
Off-label treatments
Many treatments are used despite despite the lack of phase III clinical trials characterizing their efficacy.[1] Neither inferiority nor superiority to the newer, FDA approved drugs has been clearly demonstrated, and considering their reduced price and availability in pill format, remain the current standard treatment.[1] Most of these medications affect the immune system in various ways.[48][31][55]
| Drug (brand) | Mechanism of action | Note |
|---|---|---|
| azathioprine (Imuran, Azasan) | Inhibits purine metabolism | First reported effective in 1998 and was mainstay of treatment 10+ years thereafter. Sometimes combined with steroids due to months-long onset of action.[1] |
| mycophenolate mofetil (CellCept) | Inhibits purine metabolism | Has partially replaced azathioprine due to proposed better efficacy and tolerability. Sometimes combined with steroids due to months-long onset of action.[1] |
| corticosteroid | [56] | |
| mitoxantrone | DNA synthesis/repair inhibitor | |
| methotrexate | Inhibits folate metabolism | |
| cyclophosphamide | DNA crosslinker | |
| rituximab (Rituxan) | antibody against CD20 - B cell depletion[57] | The most commonly used treatment for NMOSD today.[58] |
| intravenous immunoglobulin (IVIG) | ||
| hematopoietic stem cell transplantation (HSCT) | can be used in severe cases of NMO. Available data suggests that this procedure can reduce inflammatory activity in the short term, but a clear majority of the patients will relapse within 5 years.[59] |
It is important to note that certain immunosuppressants used to treat MS, such as interferon-β, fingolimod, natalizumab, and alemtuzumab, worsen NMO disease progression and should not be used to treat NMO.[60]
Prognosis
Normally, some measure of improvement appears in a few weeks, but residual signs and disability may persist severely sometimes.
The disease can be monophasic, i.e. a single episode with permanent remission. However, at least 85% of patients have a relapsing form of the disease with repeated attacks of transverse myelitis and/or optic neuritis. In patients with the monophasic form, the transverse myelitis and optic neuritis occur simultaneously or within days of each other. On the other hand, patients with the relapsing form are more likely to have weeks or months between the initial attacks, and to have better motor recovery after the initial transverse myelitis event. Relapses usually occur early, with about 55% of patients having a relapse in the first year and 90% in the first five years.[8]
It is possible that the relapsing form is related to the anti-AQP4+ seropositive status and the monophasic form related to its absence[61] Unlike MS, NMO rarely has a secondary progressive phase in which patients have increasing neurologic decline between attacks without remission. Instead, disabilities arise from the acute attacks.[8]
Approximately 20% of patients with monophasic NMO have permanent visual loss, and 30% have permanent paralysis in one or both legs. Among patients with relapsing NMO, 50% have paralysis or blindness within five years. In some patients (33% in one study), transverse myelitis in the cervical spinal cord resulted in respiratory failure and subsequent death. However, the spectrum of NMO has widened due to improved diagnostic criteria, and the options for treatment have improved; as a result, researchers believe these estimates will be lowered.[8]
Epidemiology
Prevalence varies by region, ranging from 0.5 to 10 per 100,000 people.[1] Unlike MS, prevalence has not been found to be related to latitude.[1] NMO is more common in women than men, with women comprising over two-thirds of patients and more than 80% of those with the relapsing form of the disease.[8]
A retrospective study found that prevalence of #Neuromyelitis optica spectrum disorders (NMOSD) was 1.5% inside a random sample of neurological patients, with a MS MS:NMOSD ratio of 42.7. Among 13 NMOSD patients, 77% had long spinal cord lesions, 38% had severe optic neuritis, and 23% had brain or brainstem lesions. Only 56% had clinically definite NMO at follow-up.[62]
NMO is more common in Asians than Caucasians. In fact, Asian optic-spinal multiple sclerosis (OSMS) (which constitutes 30% of the cases of MS in Japan) has been suggested to be identical to NMO (differences between OSMS and classic MS in Japanese patients). In the indigenous populations of tropical and subtropical regions, MS is rare, but when it appears, it often takes the form of OSMS.[63]
The majority of NMO patients have no affected relatives, and it is generally regarded as a nonfamilial condition.[8]
History
First reports on an association of spinal cord and optic nerve disorders date back to the late 18th and early 19th century.[64][65] However, only an 1870 report by Sir Thomas Clifford Allbutt created sustained interest of neurologists and ophthalmologists in this rare syndrome.[66] In 1894, Eugène Devic and his PhD student Fernand Gault described 16 patients who had lost vision in one or both eyes and within weeks developed severe spastic weakness of the limbs, loss of sensation and often bladder control. They recognized these symptoms were the result of inflammation of the optic nerve and spinal cord, respectively.[64][67][68]
In 2002, Mayo Clinic researchers identified an humoral mechanism, targeting a perivascular protein, as the culprit for NMO [16] and in 2004 an unknown specific autoantibody was found.[69] In 2005 they identified the aquaporin 4 protein as the target of the disease, and developed first in-house test to aid in the diagnosis of NMO by detection of an antibody, AQP4-IgG, in the blood.[9] First quantitative ELISA kits were developed later.[70] Later some other autoantibodies have been found in NMO AQP4-negative cases, like anti-MOG IgG, but some NMO anti-AQP4-negative cases still remain idiopathic.
Research directions
Since the discovery of AQP4 involvement, some research studies have focused on targeted treatment aimed at anti-aquaporin 4 antibodies. The most established method for antibody removal is plasmapheresis. A number of drugs are being studied: aquaporumab (non-pathogenic antibody blocker of AQP4-IgG binding), sivelestat (neutrophil elastase inhibitor), and eculizumab (complement inhibitor).[71]
There is little research into the primary causes of the anti-AQP4 auto-antibodies. It has been noticed that some cases could be paraneoplastic.[6]
In addition, several NMO variants have been discovered with antibodies other than those for AQP4, turning NMO into a heterogeneous disease. Six different patterns of damage have been reported in NMO, raising the possibility of six different types of auto-antibodies. As of 2019, only three of them are known.[72]
Research into new autoantibodies
An autoantibody (glial fibrillary acidic protein (GFAP)) was found in 2016, in transverse myelitis (LETM) and atypical NMO, leading to the concept of autoimmune GFAP astrocytopathy.[27]
Other autoantibody under research is flotillin. It has been found in seronegative NMO and some MS patients.[73]
Finally, other proteins under study are connexin 43 and anti-AQP1[74] though, as of 2015, there are only initial reports about the involvement of these proteins.[44][46]
The group AQP4+/MOG+ is very small and it can be considered a coincidence of two independent problems in the same person. Assuming these cases could be verified, five different kinds of NMO are being considered:
- NMO derived from an autoimmune channelopathy (AQP4-Ab+), around 80% of the cases
- NMO derived from an anti-MOG associated encephalomyelitis,[29] around 10% of the cases
- Connexin-43 NMO
- Aquaporin-1 associated NMO[74] which could be related to pattern III MS[75]
- Idiopathic NMO, defined by the absence of all previous antibodies
Antibody negative neuromyelitis optica
Some cases of NMO are not due to autoantibodies. They constitute an overlap between NMO and MS. Some statistical studies show that antibody negative NMO can be classified in three groups, and that this classification has a pathogenic meaning.[76]
Notable patients
See also
References
- Jarius, Sven; Paul, Friedemann; Weinshenker, Brian G.; Levy, Michael; Kim, Ho Jin; Wildemann, Brigitte (2020-10-22). "Neuromyelitis optica". Nature Reviews. Disease Primers. 6 (1): 85. doi:10.1038/s41572-020-0214-9. ISSN 2056-676X. PMID 33093467. S2CID 224825516.
- "Neuromyelitis optica - Symptoms and causes". Mayo Clinic. Retrieved 2020-08-01.
- Lana-Peixoto, Marco A.; Talim, Natália (2019-06-12). "Neuromyelitis Optica Spectrum Disorder and Anti-MOG Syndromes". Biomedicines. 7 (2): 42. doi:10.3390/biomedicines7020042. ISSN 2227-9059. PMC 6631227. PMID 31212763.
- Nasralla, Salam; Abboud, Hesham (November 2020). "Is neuromyelitis optica without AQP4-IgG a T-cell mediated disease? insights from checkpoint inhibitor immune-related adverse events". Multiple Sclerosis and Related Disorders. 46: 102451. doi:10.1016/j.msard.2020.102451. PMID 32835902.
- Pittock SJ, Lucchinetti CF (February 2016). "Neuromyelitis optica and the evolving spectrum of autoimmune aquaporin-4 channelopathies: a decade later". Annals of the New York Academy of Sciences. 1366 (1): 20–39. Bibcode:2016NYASA1366...20P. doi:10.1111/nyas.12794. PMC 4675706. PMID 26096370.
- Iorio R, Rindi G, Erra C, Damato V, Ferilli M, Sabatelli M (May 2015). "Neuromyelitis optica spectrum disorder as a paraneoplastic manifestation of lung adenocarcinoma expressing aquaporin-4". Multiple Sclerosis. 21 (6): 791–4. doi:10.1177/1352458515572241. PMID 25716881. S2CID 22763815.
- Kovacs KT, Kalluri SR, Boza-Serrano A, Deierborg T, Csepany T, Simo M, Rokusz L, Miseta A, Alcaraz N, Czirjak L, Berki T, Molnar T, Hemmer B, Illes Z (August 2016). "Change in autoantibody and cytokine responses during the evolution of neuromyelitis optica in patients with systemic lupus erythematosus: A preliminary study". Multiple Sclerosis. 22 (9): 1192–201. doi:10.1177/1352458515613165. PMID 26514978. S2CID 3808843.
- Wingerchuk DM (May 2006). "Neuromyelitis optica". International MS Journal. 13 (2): 42–50. PMID 16635421.
- Lennon VA, Kryzer TJ, Pittock SJ, Verkman AS, Hinson SR (August 2005). "IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel". The Journal of Experimental Medicine. 202 (4): 473–7. doi:10.1084/jem.20050304. PMC 2212860. PMID 16087714.
- Lucchinetti CF, Guo Y, Popescu BF, Fujihara K, Itoyama Y, Misu T (January 2014). "The pathology of an autoimmune astrocytopathy: lessons learned from neuromyelitis optica". Brain Pathology. 24 (1): 83–97. doi:10.1111/bpa.12099. PMC 3905574. PMID 24345222.
- Pittock SJ, Weinshenker BG, Lucchinetti CF, Wingerchuk DM, Corboy JR, Lennon VA (July 2006). "Neuromyelitis optica brain lesions localized at sites of high aquaporin 4 expression". Archives of Neurology. 63 (7): 964–8. doi:10.1001/archneur.63.7.964. PMID 16831965.
- Uchida T, Mori M, Uzawa A, Masuda H, Muto M, Ohtani R, Kuwabara S (July 2017). "Increased cerebrospinal fluid metalloproteinase-2 and interleukin-6 are associated with albumin quotient in neuromyelitis optica: Their possible role on blood-brain barrier disruption". Multiple Sclerosis. 23 (8): 1072–1084. doi:10.1177/1352458516672015. PMID 27682231. S2CID 4633419.
- Mørch MT, Sørensen SF, Khorooshi R, Asgari N, Owens T (April 2018). "Selective localization of IgG from cerebrospinal fluid to brain parenchyma". Journal of Neuroinflammation. 15 (1): 110. doi:10.1186/s12974-018-1159-8. PMC 5904996. PMID 29665816.
- Kowarik MC, Dzieciatkowska M, Wemlinger S, Ritchie AM, Hemmer B, Owens GP, Bennett JL (January 2015). "The cerebrospinal fluid immunoglobulin transcriptome and proteome in neuromyelitis optica reveals central nervous system-specific B cell populations". Journal of Neuroinflammation. 12: 19. doi:10.1186/s12974-015-0240-9. PMC 4323273. PMID 25626447.
- Kim SH, Kwak K, Hyun JW, Joung A, Lee SH, Choi YH, Lee JM, Kim HJ (July 2017). "Diffusion tensor imaging of normal-appearing white matter in patients with neuromyelitis optica spectrum disorder and multiple sclerosis". European Journal of Neurology. 24 (7): 966–973. doi:10.1111/ene.13321. PMID 28643955. S2CID 3941400.
- Lucchinetti CF, Mandler RN, McGavern D, Bruck W, Gleich G, Ransohoff RM, Trebst C, Weinshenker B, Wingerchuk D, Parisi JE, Lassmann H (July 2002). "A role for humoral mechanisms in the pathogenesis of Devic's neuromyelitis optica". Brain. 125 (Pt 7): 1450–61. doi:10.1093/brain/awf151. PMC 5444467. PMID 12076996.
- Ikeda K, Kiyota N, Kuroda H, Sato DK, Nishiyama S, Takahashi T, Misu T, Nakashima I, Fujihara K, Aoki M (April 2015). "Severe demyelination but no astrocytopathy in clinically definite neuromyelitis optica with anti-myelin-oligodendrocyte glycoprotein antibody". Multiple Sclerosis. 21 (5): 656–9. doi:10.1177/1352458514551455. PMID 25257613. S2CID 43699750.
- Ketelslegers IA, Van Pelt DE, Bryde S, Neuteboom RF, Catsman-Berrevoets CE, Hamann D, Hintzen RQ (October 2015). "Anti-MOG antibodies plead against MS diagnosis in an Acquired Demyelinating Syndromes cohort". Multiple Sclerosis. 21 (12): 1513–20. doi:10.1177/1352458514566666. PMID 25662345. S2CID 25321614.
- Pröbstel AK, Rudolf G, Dornmair K, Collongues N, Chanson JB, Sanderson NS, Lindberg RL, Kappos L, de Seze J, Derfuss T (March 2015). "Anti-MOG antibodies are present in a subgroup of patients with a neuromyelitis optica phenotype". Journal of Neuroinflammation. 12: 46. doi:10.1186/s12974-015-0256-1. PMC 4359547. PMID 25889963.
- Kezuka T, Usui Y, Yamakawa N, Matsunaga Y, Matsuda R, Masuda M, Utsumi H, Tanaka K, Goto H (June 2012). "Relationship between NMO-antibody and anti-MOG antibody in optic neuritis". Journal of Neuro-Ophthalmology. 32 (2): 107–10. doi:10.1097/WNO.0b013e31823c9b6c. PMID 22157536. S2CID 46667141.
- Kitley J, Waters P, Woodhall M, Leite MI, Murchison A, George J, Küker W, Chandratre S, Vincent A, Palace J (March 2014). "Neuromyelitis optica spectrum disorders with aquaporin-4 and myelin-oligodendrocyte glycoprotein antibodies: a comparative study". JAMA Neurology. 71 (3): 276–83. doi:10.1001/jamaneurol.2013.5857. PMID 24425068.
- Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG (May 2006). "Revised diagnostic criteria for neuromyelitis optica". Neurology. 66 (10): 1485–9. doi:10.1212/01.wnl.0000216139.44259.74. PMID 16717206.
- Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, Chitnis T, de Seze J, Fujihara K, Greenberg B, Jacob A, Jarius S, Lana-Peixoto M, Levy M, Simon JH, Tenembaum S, Traboulsee AL, Waters P, Wellik KE, Weinshenker BG (July 2015). "International consensus diagnostic criteria for neuromyelitis optica spectrum disorders". Neurology. 85 (2): 177–89. doi:10.1212/WNL.0000000000001729. PMC 4515040. PMID 26092914.
- Wingerchuk, Dean M.; Banwell, Brenda; Bennett, Jeffrey L.; Cabre, Philippe; Carroll, William; Chitnis, Tanuja; de Seze, Jérôme; Fujihara, Kazuo; Greenberg, Benjamin; Jacob, Anu; Jarius, Sven (2015-07-14). "International consensus diagnostic criteria for neuromyelitis optica spectrum disorders". Neurology. 85 (2): 177–189. doi:10.1212/WNL.0000000000001729. ISSN 0028-3878. PMC 4515040. PMID 26092914.
- Barkhof F, Koeller KK (February 2020). "13 Demyelinating Diseases of the CNS (Brain and Spine)". In Hodler J, Kubik-Huch RA, von Schulthess GK (eds.). Diseases of the Brain, Head and Neck, Spine 2020–2023: Diagnostic Imaging. IDKD Springer Series. Cham, Switzerland: Springer. pp. 165–176. doi:10.1007/978-3-030-38490-6_13. ISBN 978-3-030-38489-0. PMID 32119239.
- M.C. Kruer, T.K. Koch, D.N. Bourdette, D. Chabas, E. Waubant, S. Mueller, M.A. Moscarello, J. Dalmau, R.L. Woltjer, G. Adamus, NMDA RECEPTOR ENCEPHALITIS MIMICKING SERONEGATIVE NEUROMYELITIS OPTICA, Neurology, May 04, 2010; 74 (18) Clinical/Scientific Notes, DOI: https://doi.org/10.1212/WNL.0b013e3181dc1a7f
- Glial Fibrillary Acid Protein Immunoglobulin G (GFAP-IgG) Related Myelitis: Characterization and Comparison with Aquaporin-4-IgG Myelitis, Elia Sechi, P. Pearse Morris, Andrew McKeon, Sean Pittock, Shannon Hinson, Brian Weinshenker, Allen J. Aksamit, Evan A. Jolliffe, Anastasia Zekeridou, Dean Wingerchuk, Eoin P. Flanagan, Neurology Apr 2018, 90 (15 Supplement) S13.006
- Kun Jia et al., Anti-neurofascin-155 antibody-positive neuromyelitis optica spectrum disorders, Journal of Neurological Sciences, January 16, 2019, DOI: https://doi.org/10.1016/j.jns.2019.01.024
- Spadaro M, Gerdes LA, Mayer MC, Ertl-Wagner B, Laurent S, Krumbholz M, Breithaupt C, Högen T, Straube A, Giese A, Hohlfeld R, Lassmann H, Meinl E, Kümpfel T (March 2015). "Histopathology and clinical course of MOG-antibody-associated encephalomyelitis". Annals of Clinical and Translational Neurology. 2 (3): 295–301. doi:10.1002/acn3.164. PMC 4369279. PMID 25815356.
- Li Y, Xie P, Lv F, Mu J, Li Q, Yang Q, Hu M, Tang H, Yi J (October 2008). "Brain magnetic resonance imaging abnormalities in neuromyelitis optica". Acta Neurologica Scandinavica. 118 (4): 218–25. doi:10.1111/j.1600-0404.2008.01012.x. PMID 18384459. S2CID 22270592.
- Wingerchuk, Dean (2006). "Neuromyelitis Optica (Devic's Syndrome)" (PDF). 2006 Rare Neuroimmunologic Disorders Symposium. Archived from the original (PDF) on 2006-09-25. Retrieved 2007-01-05.
- Fujihara K, Leite MI (June 2013). "Seronegative NMO: a sensitive AQP4 antibody test clarifies clinical features and next challenges". Neurology. 80 (24): 2176–7. doi:10.1212/WNL.0b013e318296ea22. PMID 23658387. S2CID 207123279.
- Marignier R, Bernard-Valnet R, Giraudon P, Collongues N, Papeix C, Zéphir H, Cavillon G, Rogemond V, Casey R, Frangoulis B, De Sèze J, Vukusic S, Honnorat J, Confavreux C (June 2013). "Aquaporin-4 antibody-negative neuromyelitis optica: distinct assay sensitivity-dependent entity". Neurology. 80 (24): 2194–200. doi:10.1212/WNL.0b013e318296e917. PMID 23658379. S2CID 25662273.
- Bergamaschi R, Tonietti S, Franciotta D, Candeloro E, Tavazzi E, Piccolo G, Romani A, Cosi V (February 2004). "Oligoclonal bands in Devic's neuromyelitis optica and multiple sclerosis: differences in repeated cerebrospinal fluid examinations". Multiple Sclerosis. 10 (1): 2–4. doi:10.1191/1352458504ms988oa. PMID 14760945. S2CID 11730134.
- Komatsu J, Sakai K, Nakada M, Iwasa K, Yamada M (August 2017). "Long spinal cord lesions in a patient with pathologically proven multiple sclerosis". Journal of Clinical Neuroscience. 42: 106–108. doi:10.1016/j.jocn.2017.03.022. PMID 28465080. S2CID 3443914.
- Arru G, Sechi E, Mariotto S, Farinazzo A, Mancinelli C, Alberti D, Ferrari S, Gajofatto A, Capra R, Monaco S, Deiana GA, Caggiu E, Mameli G, Sechi LA, Sechi GP (2017). "Antibody response against HERV-W env surface peptides differentiates multiple sclerosis and neuromyelitis optica spectrum disorder". Multiple Sclerosis Journal – Experimental, Translational and Clinical. 3 (4): 2055217317742425. doi:10.1177/2055217317742425. PMC 5703109. PMID 29204291.
- Jurynczyk M, Probert F, Yeo T, Tackley G, Claridge TD, Cavey A, Woodhall MR, Arora S, Winkler T, Schiffer E, Vincent A, DeLuca G, Sibson NR, Isabel Leite M, Waters P, Anthony DC, Palace J (December 2017). "Metabolomics reveals distinct, antibody-independent, molecular signatures of MS, AQP4-antibody and MOG-antibody disease". Acta Neuropathologica Communications. 5 (1): 95. doi:10.1186/s40478-017-0495-8. PMC 5718082. PMID 29208041.
- Pearce JM (November 2005). "Neuromyelitis optica". Spinal Cord. 43 (11): 631–4. doi:10.1038/sj.sc.3101758. PMID 15968305.
- Lassmann H, Brück W, Lucchinetti C (March 2001). "Heterogeneity of multiple sclerosis pathogenesis: implications for diagnosis and therapy". Trends in Molecular Medicine. 7 (3): 115–21. doi:10.1016/s1471-4914(00)01909-2. PMID 11286782.
- Schneider R (2013). "Autoantibodies to Potassium Channel KIR4.1 in Multiple Sclerosis". Frontiers in Neurology. 4: 125. doi:10.3389/fneur.2013.00125. PMC 3759297. PMID 24032025.
- Kurosawa K, Fujihara K (November 2014). "[Clinical concept, etiology and pathology of neuromyelitis optica]". Nihon Rinsho. Japanese Journal of Clinical Medicine. 72 (11): 1897–902. PMID 25518368.
- Harmel J, Ringelstein M, Ingwersen J, Mathys C, Goebels N, Hartung HP, Jarius S, Aktas O (December 2014). "Interferon-β-related tumefactive brain lesion in a Caucasian patient with neuromyelitis optica and clinical stabilization with tocilizumab". BMC Neurology. 14: 247. doi:10.1186/s12883-014-0247-3. PMC 4301061. PMID 25516429.
- Alexander B. Ramos et al., A case of NMO spectrum disorder presenting with undiagnosed Sjogren's syndrome and a single, atypical tumefactive lesion: A clinical conundrum, Neur. Sciences, December 15, 2017Volume 383, Pages 216–218, DOI: https://doi.org/10.1016/j.jns.2017.10.036
- Masaki K, Suzuki SO, Matsushita T, Matsuoka T, Imamura S, Yamasaki R, Suzuki M, Suenaga T, Iwaki T, Kira J (2013). "Connexin 43 astrocytopathy linked to rapidly progressive multiple sclerosis and neuromyelitis optica". PLOS ONE. 8 (8): e72919. Bibcode:2013PLoSO...872919M. doi:10.1371/journal.pone.0072919. PMC 3749992. PMID 23991165.
- Fujihara K, Sato DK (October 2013). "AQP4 antibody serostatus: Is its luster being lost in the management and pathogenesis of NMO?". Neurology. 81 (14): 1186–8. doi:10.1212/WNL.0b013e3182a6cc23. PMID 23997154. S2CID 35351168.
- Masaki K (October 2015). "Early disruption of glial communication via connexin gap junction in multiple sclerosis, Baló's disease and neuromyelitis optica". Neuropathology. 35 (5): 469–80. doi:10.1111/neup.12211. PMID 26016402. S2CID 6371457.
- "Neuromyelitis optica - Diagnosis and treatment - Mayo Clinic". www.mayoclinic.org. Retrieved 2020-08-03.
- Kowarik, Markus C.; Soltys, John; Bennett, Jeffrey L. (March 2014). "The Treatment of Neuromyelitis Optica". Journal of Neuro-Ophthalmology. 34 (1): 70–82. doi:10.1097/WNO.0000000000000102. ISSN 1070-8022. PMC 4208473. PMID 24531318.
- Wallach, AI; Tremblay, M; Kister, I (February 2021). "Advances in the Treatment of Neuromyelitis Optica Spectrum Disorder". Neurologic clinics. 39 (1): 35–49. doi:10.1016/j.ncl.2020.09.003. PMID 33223088.
- Kimbrough, Dorlan J.; Fujihara, Kazuo; Jacob, Anu; Lana-Peixoto, Marco A.; Isabel Leite, Maria; Levy, Michael; Marignier, Romain; Nakashima, Ichiro; Palace, Jacqueline; de Seze, Jérôme; Stuve, Olaf; Tenembaum, Silvia N.; Traboulsee, Anthony; Waubant, Emmanuelle; Weinshenker, Brian G.; Wingerchuk, Dean M. (October 2012). "Treatment of neuromyelitis optica: Review and recommendations". Multiple Sclerosis and Related Disorders. 1 (4): 180–187. doi:10.1016/j.msard.2012.06.002. PMC 3926208. PMID 24555176.
- "FDA approves first treatment for neuromyelitis optica spectrum disorder, a rare autoimmune disease of the central nervous system". U.S. Food and Drug Administration (FDA) (Press release). 27 June 2019. Retrieved 28 June 2019.
- "FDA Approves New Therapy for Rare Disease Affecting Optic Nerve, Spinal Cord". U.S. Food and Drug Administration (FDA) (Press release). 2020-06-11. Retrieved 2020-06-21.
- "Enspryng (satralizumab)". Roche. Retrieved 9 August 2020.
- "FDA Approves Treatment for Rare Disease Affecting Optic Nerves, Spinal Cord". U.S. Food and Drug Administration (FDA). 17 August 2020. Retrieved 17 August 2020.
- Weinstock-Guttman B, Ramanathan M, Lincoff N, Napoli SQ, Sharma J, Feichter J, Bakshi R (July 2006). "Study of mitoxantrone for the treatment of recurrent neuromyelitis optica (Devic disease)". Archives of Neurology. 63 (7): 957–63. doi:10.1001/archneur.63.7.957. PMID 16831964.
- Watanabe S, Misu T, Miyazawa I, Nakashima I, Shiga Y, Fujihara K, Itoyama Y (September 2007). "Low-dose corticosteroids reduce relapses in neuromyelitis optica: a retrospective analysis". Multiple Sclerosis. 13 (8): 968–74. doi:10.1177/1352458507077189. PMID 17623727. S2CID 6308153.
- Matiello M, Jacob A, Wingerchuk DM, Weinshenker BG (June 2007). "Neuromyelitis optica". Current Opinion in Neurology. 20 (3): 255–60. doi:10.1097/WCO.0b013e32814f1c6b. PMID 17495617. S2CID 21483082.
- Evangelopoulos ME, Andreadou E, Koutsis G, Koutoulidis V, Anagnostouli M, Katsika P, Evangelopoulos DS, Evdokimidis I, Kilidireas C (January 2017). "Treatment of neuromyelitis optica and neuromyelitis optica spectrum disorders with rituximab using a maintenance treatment regimen and close CD19 B cell monitoring. A six-year follow-up". Journal of the Neurological Sciences. 372: 92–96. doi:10.1016/j.jns.2016.11.016. PMID 28017256. S2CID 206291987.
- Burman J, Tolf A, Hägglund H, Askmark H (February 2018). "Autologous haematopoietic stem cell transplantation for neurological diseases". Journal of Neurology, Neurosurgery, and Psychiatry. 89 (2): 147–155. doi:10.1136/jnnp-2017-316271. PMC 5800332. PMID 28866625.
- Baghbanian, Seyed Mohammad; Asgari, Nasrin; Sahraian, Mohammad Ali; Moghadasi, Abdorreza Naser (May 2018). "A comparison of pediatric and adult neuromyelitis optica spectrum disorders: A review of clinical manifestation, diagnosis, and treatment". Journal of the Neurological Sciences. 388: 222–231. doi:10.1016/j.jns.2018.02.028. PMID 29478727. S2CID 3543215.
- Ketelslegers IA, Modderman PW, Vennegoor A, Killestein J, Hamann D, Hintzen RQ (December 2011). "Antibodies against aquaporin-4 in neuromyelitis optica: distinction between recurrent and monophasic patients". Multiple Sclerosis. 17 (12): 1527–30. doi:10.1177/1352458511412995. PMID 21828202. S2CID 206698444.
- Bizzoco E, Lolli F, Repice AM, Hakiki B, Falcini M, Barilaro A, Taiuti R, Siracusa G, Amato MP, Biagioli T, Lori S, Moretti M, Vinattieri A, Nencini P, Massacesi L, Matà S (November 2009). "Prevalence of neuromyelitis optica spectrum disorder and phenotype distribution". Journal of Neurology. 256 (11): 1891–8. doi:10.1007/s00415-009-5171-x. PMID 19479168. S2CID 26122372.
- Cabre P, Signate A, Olindo S, Merle H, Caparros-Lefebvre D, Béra O, Smadja D (December 2005). "Role of return migration in the emergence of multiple sclerosis in the French West Indies". Brain. 128 (Pt 12): 2899–910. doi:10.1093/brain/awh624. PMID 16183661.
- Jarius S, Wildemann B (January 2013). "The history of neuromyelitis optica". Journal of Neuroinflammation. 10 (1): 8. doi:10.1186/1742-2094-10-8. PMC 3599417. PMID 23320783.
- Jarius S, Wildemann B (November 2018). "The history of neuromyelitis optica. Part 2: 'Spinal amaurosis', or how it all began". Journal of Neuroinflammation. 16 (1): 280. doi:10.1186/s12974-019-1594-1. PMC 6935230. PMID 31883522.
- Jarius S, Wildemann B (January 2013). "On the contribution of Thomas Clifford Allbutt, F.R.S., to the early history of neuromyelitis optica". Journal of Neurology. 260 (1): 100–4. doi:10.1007/s00415-012-6594-3. PMID 22782261. S2CID 23878209.
- Devic E (1894). "Myélite subaiguë compliquée de névrite optique" [Subacute myelitis complicated with optic neuritis]. Bull Med (in French). 8: 1033.
- T. Jock Murray (2005). Multiple Sclerosis: The History of a Disease. New York: Demos Medical Publishing. ISBN 978-1-888799-80-4.
- Lennon VA, Wingerchuk DM, Kryzer TJ, Pittock SJ, Lucchinetti CF, Fujihara K, Nakashima I, Weinshenker BG (2004). "A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis". Lancet. 364 (9451): 2106–12. doi:10.1016/S0140-6736(04)17551-X. PMID 15589308. S2CID 29316257.
- Isobe, Noriko; Yonekawa, Tomomi; Matsushita, Takuya; Kawano, Yuji; Masaki, Katsuhisa; Yoshimura, Satoshi; Fichna, Jakub; Chen, Shu; Furmaniak, Jadwiga; Smith, Bernard Rees; Kira, Jun-Ichi (2012). "Quantitative assays for anti-aquaporin-4 antibody with subclass analysis in neuromyelitis optica". Multiple Sclerosis Journal. 18 (11): 1541–1551. doi:10.1177/1352458512443917. PMID 22526930. S2CID 206699115.
- Papadopoulos MC, Verkman AS (June 2012). "Aquaporin 4 and neuromyelitis optica". The Lancet. Neurology. 11 (6): 535–44. doi:10.1016/S1474-4422(12)70133-3. PMC 3678971. PMID 22608667.
- Misu T, Höftberger R, Fujihara K, Wimmer I, Takai Y, Nishiyama S, Nakashima I, Konno H, Bradl M, Garzuly F, Itoyama Y, Aoki M, Lassmann H (June 2013). "Presence of six different lesion types suggests diverse mechanisms of tissue injury in neuromyelitis optica". Acta Neuropathologica. 125 (6): 815–27. doi:10.1007/s00401-013-1116-7. PMC 3661909. PMID 23579868.
- Hahn S, Trendelenburg G, Scharf M, Denno Y, Brakopp S, Teegen B, Probst C, Wandinger KP, Buttmann M, Haarmann A, Szabados F, Vom Dahl M, Kümpfel T, Eichhorn P, Gold H, Paul F, Jarius S, Melzer N, Stöcker W, Komorowski L (June 2017). "Identification of the flotillin-1/2 heterocomplex as a target of autoantibodies in bona fide multiple sclerosis". Journal of Neuroinflammation. 14 (1): 123. doi:10.1186/s12974-017-0900-z. PMC 5481867. PMID 28645295.
- Tzartos JS, Stergiou C, Kilidireas K, Zisimopoulou P, Thomaidis T, Tzartos SJ (2013). "Anti-aquaporin-1 autoantibodies in patients with neuromyelitis optica spectrum disorders". PLOS ONE. 8 (9): e74773. Bibcode:2013PLoSO...874773T. doi:10.1371/journal.pone.0074773. PMC 3781161. PMID 24086369.CS1 maint: multiple names: authors list (link)
- Stork, Lidia; Ellenberger, David; Ruprecht, Klemens; Reindl, Markus; Beißbarth, Tim; Friede, Tim; Kümpfel, Tania; Gerdes, Lisa A.; Gloth, Mareike; Liman, Thomas; Paul, Friedemann; Brück, Wolfgang; Metz, Imke (2020). "Antibody signatures in patients with histopathologically defined multiple sclerosis patterns". Acta Neuropathologica. 139 (3): 547–564. doi:10.1007/s00401-019-02120-x. PMC 7035238. PMID 31950335.
- Tianrong Yeo, Fay Probert, Maciej Jurynczyk, Megan Sealey, Ana Cavey, Timothy D.W. Claridge, Mark Woodhall, Patrick Waters, Maria Isabel Leite, Daniel C. Anthony, Jacqueline Palace, Classifying the antibody-negative NMO syndromes, Clinical, imaging, and metabolomic modeling, October 28, 2019, DOI: https://doi.org/10.1212/NXI.0000000000000626
External links
| Classification | |
|---|---|
| External resources |
