Lead tetrafluoride
Lead tetrafluoride is a compound of lead and fluorine. The yellow solid (melting point 600 °C) is the only room-temperature stable tetrahalide of lead.[5] Lead tetrafluoride is isostructural with tin(IV) fluoride and contains planar layers of octahedrally coordinated lead, where the octahedra share four corners and there are two terminal, unshared, fluorine atoms trans to one another.[6]
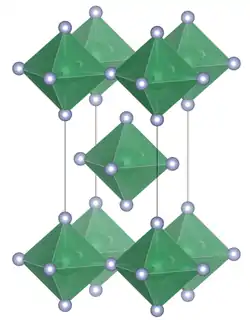 Unit cell of lead tetrafluoride | |
| Names | |
|---|---|
| IUPAC name
Tetrafluoroplumbane | |
| Other names
Lead(IV) fluoride Lead tetrafluoride Tetrafluoridolead | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.029.102 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| PbF4 | |
| Molar mass | 283.194 g/mol [1] |
| Appearance | white to beige crystals [2] |
| Density | 6.7 g/cm3 [3] |
| Melting point | 600 °C (1,112 °F; 873 K)[4] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
References
- https://pubchem.ncbi.nlm.nih.gov/compound/Lead_IV__fluoride
- http://www.chemicalbook.com/ChemicalProductProperty_EN_CB5727780.htm
- http://www.chemicalbook.com/ChemicalProductProperty_EN_CB5727780.htm
- http://www.chemicalbook.com/ChemicalProductProperty_EN_CB5727780.htm
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 375–376, 381–382. ISBN 978-0-08-037941-8.
- Inorganic Chemistry [Paperback],2d Edition, Housecroft, Sharpe,2004, Pearson Education ISBN 0130399132, ISBN 978-0130399137
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.