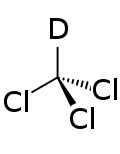
Deuterated chloroform
Deuterated chloroform (CDCl3), also known as chloroform-d, is an isotopically enriched form of chloroform (CHCl3) in which most its hydrogen atoms consist of the heavier nuclide deuterium (heavy hydrogen) (D = 2H) rather than the natural isotopic mixture in which protium (1H) is predominant.[2] Deuterated chloroform is by far the most common solvent used in NMR spectroscopy.[3] While dichloromethane and chloroform (trichloromethane) are both commonly used solvents that dissolve a wide range of other organic compounds, deuterated chloroform is more easily produced and much less expensive than deuterated dichloromethane.[4] In addition, it is chemically unreactive and unlikely to exchange its deuterium with its solute, and its low boiling point allows for easy sample recovery.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
trichloro(deuterio)methane[1] | |||
| Other names
Chloroform-d Deuterochloroform | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 1697633 | |||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.011.585 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
| UN number | 1888 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CDCl3 | |||
| Molar mass | 120.384 g mol−1 | ||
| Density | 1.500 g cm−3 | ||
| Melting point | −64 °C (−83 °F; 209 K) | ||
| Boiling point | 61 °C (142 °F; 334 K) | ||
| Hazards | |||
| GHS pictograms |    | ||
| GHS Signal word | Danger | ||
| H302, H315, H319, H331, H336, H351, H361, H372, H373 | |||
| P201, P202, P260, P261, P264, P270, P271, P280, P281, P301+312, P302+352, P304+340, P305+351+338, P308+313, P311, P312, P314, P321, P330, P332+313, P337+313, P362, P403+233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Related compounds |
Chloroform | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
The properties of CDCl3 are virtually identical to those of regular chloroform, although biologically, it is slightly less toxic to the liver than CHCl3, due to its C–D bond, which is stronger than a C–H bond, making it somewhat less prone to form the destructive trichloromethyl radical (•CCl3).[5]
NMR solvent
In proton NMR spectroscopy, deuterated solvent (enriched to >99% deuterium) must be used to avoid recording a large interfering signal or signals from the proton(s) (i.e., hydrogen-1) present in the solvent itself. If nondeuterated chloroform (containing a full equivalent of protium) were used as solvent, the solvent signal would almost certainly overwhelm and obscure any nearby analyte signals. In addition, modern instruments usually require the presence of deuterated solvent, as the field frequency is locked using the deuterium signal of the solvent to prevent frequency drift. Commercial chloroform-d does, however, still contain a small amount (0.2% or less) of non-deuterated chloroform; this results in a small singlet at 7.26 ppm, known as the residual solvent peak, which is frequently used as an internal chemical shift reference.
In carbon-13 NMR spectroscopy, the sole carbon in deuterated chloroform shows a triplet at a chemical shift of 77.16 ppm with the three peaks being about equal size, resulting from splitting by spin coupling to the attached spin-1 deuterium atom (CHCl3 has a chemical shift of 77.36 ppm).[4]
It reacts photochemically with oxygen to form phosgene and hydrogen chloride. Therefore, more expensive alternatives like dichloromethane-d2 or benzene-d6 must be used if the analyte is expected to be highly acid-sensitive. To slow this process and reduce the acidity of the solvent, chloroform-d is stored in brown-tinted bottles, often over a small amount of a neutralizing base like potassium carbonate.
Hazards
Like nondeuterated chloroform, chloroform-d is hepatotoxic and likely to be carcinogenic. In addition, exposure to light and oxygen results in the formation of highly toxic phosgene.
References
- https://pubchem.ncbi.nlm.nih.gov/compound/Chloroform-D
- Since non-enriched hydrogen is mostly protium (99.85%), with only a trace of deuterium (0.15%), this enrichment in deuterium is colloquially, though somewhat inaccurately, described as "replacement" of hydrogen ("H") with deuterium ("D").
- Fulmer, Gregory R.; Miller, Alexander J. M.; Sherden, Nathaniel H.; Gottlieb, Hugo E.; Nudelman, Abraham; Stoltz, Brian M.; Bercaw, John E.; Goldberg, Karen I. (2010). "NMR Chemical Shifts of Trace Impurities: Common Laboratory Solvents, Organics, and Gases in Deuterated Solvents Relevant to the Organometallic Chemist" (PDF). Organometallics. 29 (9): 2176–2179. doi:10.1021/om100106e.
- The Theory of NMR – Solvents for NMR spectroscopy
- Goldstein, Robin S. (2013). Toxic interactions. Hewitt, William R., Hook, Jerry B. Burlington: Elsevier Science. ISBN 978-1-4832-6970-2. OCLC 896796140.