Sodium metasilicate
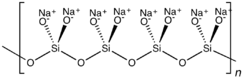
Sodium metasilicate is the chemical substance with formula Na
2SiO
3, which is the main component of commercial sodium silicate solutions. It is an ionic compound consisting of sodium cations Na+
and the polymeric metasilicate anions [–SiO2−
3–]n. It is a colorless crystalline hygroscopic and deliquescent solid, soluble in water (giving an alkaline solution) but not in alcohols.[1]
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Sodium metasilicate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| Abbreviations | E550 |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.027.193 |
| EC Number |
|
| MeSH | Sodium+metasilicate |
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| UN number | 1759 3253 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Na2SiO3 | |
| Molar mass | 122.062 g·mol−1 |
| Appearance | White crystals |
| Density | 2.61 g/cm3 |
| Melting point | 1,088 °C (1,990 °F; 1,361 K) |
| 22.2 g/100 ml (25 °C) 160.6 g/100 ml (80 °C) | |
| Solubility | insoluble in alcohol |
Refractive index (nD) |
1.52 |
| Thermochemistry | |
Heat capacity (C) |
111.8 J/(K·mol) |
Std molar entropy (S |
113.71 J/(K·mol) |
Std enthalpy of formation (ΔfH⦵298) |
−1561.43 kJ/mol |
Gibbs free energy (ΔfG˚) |
−1427 kJ/mol |
| Hazards | |
| Safety data sheet | Avantor Performance Materials |
| GHS pictograms |   |
| GHS Signal word | Danger |
| H302, H314, H315, H318, H319, H335 | |
| P260, P261, P264, P270, P271, P280, P301+312, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P312, P321, P330, P332+313, P337+313, P362, P363, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1153 (rat, oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation and properties
The anhydrous compound can be prepared by fusing silicon dioxide SiO
2 (silica, quartz) with sodium oxide Na
2O in 1:1 molar ratio.[2]
The compound crystallizes from solution as various hydrates, such as
- pentahydrate Na
2SiO
3·5H
2O (CAS 10213-79-3, EC 229-912-9, PubChem 57652358) - nonahydrate Na
2SiO
3·9H
2O (CAS 13517-24-3, EC 229-912-9, PubChem 57654617)[3]
Structure
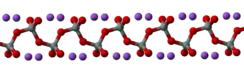
In the anhydrous solid, the metasilicate anion is actually polymeric, consisting of corner-shared {SiO4} tetrahedra, and not a discrete SiO32− ion.[4]
In addition to the anhydrous form, there are hydrates with the formula Na2SiO3·nH2O (where n = 5, 6, 8, 9), which contain the discrete, approximately tetrahedral anion SiO2(OH)22− with water of hydration. For example, the commercially available sodium silicate pentahydrate Na2SiO3·5H2O is formulated as Na2SiO2(OH)2·4H2O, and the nonahydrate Na2SiO3·9H2O is formulated as Na2SiO2(OH)2·8H2O.[5] The pentahydrate and nonahydrate forms have their own CAS Numbers, 10213-79-3 and 13517-24-3 respectively.
Uses
Sodium Metasilicate reacts with acids to produce silica gel.[6]
- Cements and Binders - dehydrated sodium metasilicate forms cement or binding agent.
- Pulp and Par - sizing agent and buffer/stabilizing agent when mixed with hydrogen peroxide.
- Soaps and Detergents - as an emulsifying and suspension agent.
- Automotive applications - decommissioning of old engines (CARS program), cooling system sealant, exhaust repair.
- Egg Preservative - seals eggs increasing shelf life.
- Crafts - forms "stalagmites" by reacting with and precipitating metal ions. Also used as a glue called "soluble glass".
References
- Chemical Book: "Sodium metasilicate". Accessed on 2018-05-13.
- J. F. Schairer and N. L. Bowen (1956): "The system Na
2O—Al
2O
3—SiO
2". American Journal of Science, volume 254, issue 3, pages 129-195 doi:10.2475/ajs.254.3.129 - M. F. Bechtold (1955): "Polymerization and Properties of Dilute Aqueous Silicic Acid from Cation Exchange" Journal of Physical Chemistry, volume 59, issue 6, pages 532–541. doi:10.1021/j150528a013
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- https://sciencing.com/uses-sodium-metasilicate-5447484.html
