Hexachloroacetone
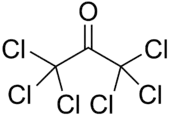
Hexachloroacetone is an organic compound with the formula (Cl3C)2CO. It is also called hexachloropropanone or perchloroacetone. Numbers indicating the position of the chlorine-atoms are generally omitted as all the possible positions are substituted with chlorine. It is a colorless liquid, slightly soluble in water.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
1,1,1,3,3,3-hexachloropropanone | |
| Other names
perchloroacetone HCA | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.754 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2661 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3Cl6O | |
| Molar mass | 264.75 g/mol |
| Density | 1.7434 g/cm3[1] |
| Melting point | −2 °C (28 °F; 271 K) |
| Boiling point | 204 °C (399 °F; 477 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The main use of hexachloroacetone is as a pesticide. For the use of hexachloroacetone in the preparation of a novel insect repellent see Perkow reaction. The industrial route to hexafluoroacetone involves treatment of hexachloroacetone with HF:[2]
- (CCl3)2CO + 6 HF → (CF3)2CO + 6 HCl
See also
References
- CRC Handbook of Chemistry and Physics, Internet Version, CRC Press (2005)
- Günter Siegemund, Werner Schwertfeger, Andrew Feiring, Bruce Smart, Fred Behr, Herward Vogel, Blaine McKusick (2002). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349. ISBN 3527306730.CS1 maint: uses authors parameter (link)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.