H3K14ac
H3K14ac is an epigenetic modification to the DNA packaging protein Histone H3. It is a mark that indicates the acetylation at the 14th lysine residue of the histone H3 protein.
H3K14ac has not been widely studied partly due to previous lack of commercially available antibody. H3K9ac and H3K14ac have been shown to be part of the active promoter state. They are also present over bivalent promoters and active enhancers. H3K14ac is also enriched over a subset of inactive promoters.
The Tudor domain of the H3K9 methyltransferase SETDB1 binds to methylated H3 with both K14 acetylation and K9 methylation. SETDB1 silences retroviruses and gene regulation.
Lysine acetylation and deacetylation
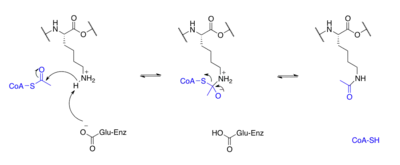
Proteins are typically acetylated on lysine residues and this reaction relies on acetyl-coenzyme A as the acetyl group donor. In histone acetylation and deacetylation, histone proteins are acetylated and deacetylated on lysine residues in the N-terminal tail as part of gene regulation. Typically, these reactions are catalyzed by enzymes with histone acetyltransferase (HAT) or histone deacetylase (HDAC) activity, although HATs and HDACs can modify the acetylation status of non-histone proteins as well.[1]
The regulation of transcription factors, effector proteins, molecular chaperones, and cytoskeletal proteins by acetylation and deacetylation is a significant post-translational regulatory mechanism[2] These regulatory mechanisms are analogous to phosphorylation and dephosphorylation by the action of kinases and phosphatases. Not only can the acetylation state of a protein modify its activity but there has been recent suggestion that this post-translational modification may also crosstalk with phosphorylation, methylation, ubiquitination, sumoylation, and others for dynamic control of cellular signaling.[3][4][5]
In the field of epigenetics, histone acetylation (and deacetylation) have been shown to be important mechanisms in the regulation of gene transcription. Histones, however, are not the only proteins regulated by posttranslational acetylation.
Nomenclature
H3K14acindicates acetylation of lysine 14on histone H3 protein subunit: [6]
| Abbr. | Meaning |
| H3 | H3 family of histones |
| K | standard abbreviation for lysine |
| 14 | position of amino acid residue (counting from N-terminus) |
| ac | acetyl group |
Histone modifications
The genomic DNA of eukaryotic cells is wrapped around special protein molecules known as histones. The complexes formed by the looping of the DNA are known as chromatin. The basic structural unit of chromatin is the nucleosome: this consists of the core octamer of histones (H2A, H2B, H3 and H4) as well as a linker histone and about 180 base pairs of DNA. These core histones are rich in lysine and arginine residues. The carboxyl (C) terminal end of these histones contribute to histone-histone interactions, as well as histone-DNA interactions. The amino (N) terminal charged tails are the site of the post-translational modifications, such as the one seen in H3K36me3.[7][8]
Epigenetic implications
The post-translational modification of histone tails by either histone modifying complexes or chromatin remodelling complexes are interpreted by the cell and lead to complex, combinatorial transcriptional output. It is thought that a Histone code dictates the expression of genes by a complex interaction between the histones in a particular region.[9] The current understanding and interpretation of histones comes from two large scale projects: ENCODE and the Epigenomic roadmap.[10] The purpose of the epigenomic study was to investigate epigenetic changes across the entire genome. This led to chromatin states which define genomic regions by grouping the interactions of different proteins and/or histone modifications together. Chromatin states were investigated in Drosophila cells by looking at the binding location of proteins in the genome. Use of ChIP-sequencing revealed regions in the genome characterised by different banding.[11] Different developmental stages were profiled in Drosophila as well, an emphasis was placed on histone modification relevance.[12] A look in to the data obtained led to the definition of chromatin states based on histone modifications.[13]
The human genome was annotated with chromatin states. These annotated states can be used as new ways to annotate a genome independently of the underlying genome sequence. This independence from the DNA sequence enforces the epigenetic nature of histone modifications. Chromatin states are also useful in identifying regulatory elements that have no defined sequence, such as enhancers. This additional level of annotation allows for a deeper understanding of cell specific gene regulation.[14]
H3K14ac
H3K14ac has not been widely studied partly due to previous lack of commercially available antibody. H3K9ac and H3K14ac have been shown to be part of the active promoter state. They are also present over bivalent promoters and active enhancers. H3K14ac is also enriched over a subset of inactive promoters.[15]
The Triple Tudor domain of the H3K9 methyltransferase SETDB1 binds to methylated H3 with both K14 acetylation and K9 methylation. SETDB1 silences retroviruses and gene regulation.[16]
Methods
The histone mark acetylation can be detected in a variety of ways:
1. Chromatin Immunoprecipitation Sequencing (ChIP-sequencing) measures the amount of DNA enrichment once bound to a targeted protein and immunoprecipitated. It results in good optimization and is used in vivo to reveal DNA-protein binding occurring in cells. ChIP-Seq can be used to identify and quantify various DNA fragments for different histone modifications along a genomic region.[17]
2. Micrococcal Nuclease sequencing (MNase-seq) is used to investigate regions that are bound by well positioned nucleosomes. Use of the micrococcal nuclease enzyme is employed to identify nucleosome positioning. Well positioned nucleosomes are seen to have enrichment of sequences.[18]
3. Assay for transposase accessible chromatin sequencing (ATAC-seq) is used to look in to regions that are nucleosome free (open chromatin). It uses hyperactive Tn5 transposon to highlight nucleosome localisation.[19][20][21]
See also
References
- Sadoul K, Boyault C, Pabion M, Khochbin S (2008). "Regulation of protein turnover by acetyltransferases and deacetylases". Biochimie. 90 (2): 306–12. doi:10.1016/j.biochi.2007.06.009. PMID 17681659.
- Glozak MA, Sengupta N, Zhang X, Seto E (2005). "Acetylation and deacetylation of non-histone proteins". Gene. 363: 15–23. doi:10.1016/j.gene.2005.09.010. PMID 16289629.
- Yang XJ, Seto E (2008). "Lysine acetylation: codified crosstalk with other posttranslational modifications". Mol. Cell. 31 (4): 449–61. doi:10.1016/j.molcel.2008.07.002. PMC 2551738. PMID 18722172.
- Eddé B, Denoulet P, de Néchaud B, Koulakoff A, Berwald-Netter Y, Gros F (1989). "Posttranslational modifications of tubulin in cultured mouse brain neurons and astroglia". Biol. Cell. 65 (2): 109–117. doi:10.1016/0248-4900(89)90018-x. PMID 2736326.
- Maruta H, Greer K, Rosenbaum JL (1986). "The acetylation of alpha-tubulin and its relationship to the assembly and disassembly of microtubules". J. Cell Biol. 103 (2): 571–579. doi:10.1083/jcb.103.2.571. PMC 2113826. PMID 3733880.
- Huang, Suming; Litt, Michael D.; Ann Blakey, C. (2015-11-30). Epigenetic Gene Expression and Regulation. pp. 21–38. ISBN 9780127999586.
- Ruthenburg AJ, Li H, Patel DJ, Allis CD (December 2007). "Multivalent engagement of chromatin modifications by linked binding modules". Nature Reviews. Molecular Cell Biology. 8 (12): 983–94. doi:10.1038/nrm2298. PMC 4690530. PMID 18037899.
- Kouzarides T (February 2007). "Chromatin modifications and their function". Cell. 128 (4): 693–705. doi:10.1016/j.cell.2007.02.005. PMID 17320507.
- Jenuwein T, Allis CD (August 2001). "Translating the histone code". Science. 293 (5532): 1074–80. doi:10.1126/science.1063127. PMID 11498575.
- Birney E, Stamatoyannopoulos JA, Dutta A, Guigó R, Gingeras TR, Margulies EH, et al. (The ENCODE Project Consortium) (June 2007). "Identification and analysis of functional elements in 1% of the human genome by the ENCODE pilot project". Nature. 447 (7146): 799–816. Bibcode:2007Natur.447..799B. doi:10.1038/nature05874. PMC 2212820. PMID 17571346.
- Filion GJ, van Bemmel JG, Braunschweig U, Talhout W, Kind J, Ward LD, Brugman W, de Castro IJ, Kerkhoven RM, Bussemaker HJ, van Steensel B (October 2010). "Systematic protein location mapping reveals five principal chromatin types in Drosophila cells". Cell. 143 (2): 212–24. doi:10.1016/j.cell.2010.09.009. PMC 3119929. PMID 20888037.
- Roy S, Ernst J, Kharchenko PV, Kheradpour P, Negre N, Eaton ML, et al. (modENCODE Consortium) (December 2010). "Identification of functional elements and regulatory circuits by Drosophila modENCODE". Science. 330 (6012): 1787–97. Bibcode:2010Sci...330.1787R. doi:10.1126/science.1198374. PMC 3192495. PMID 21177974.
- Kharchenko PV, Alekseyenko AA, Schwartz YB, Minoda A, Riddle NC, Ernst J, et al. (March 2011). "Comprehensive analysis of the chromatin landscape in Drosophila melanogaster". Nature. 471 (7339): 480–5. Bibcode:2011Natur.471..480K. doi:10.1038/nature09725. PMC 3109908. PMID 21179089.
- Kundaje A, Meuleman W, Ernst J, Bilenky M, Yen A, Heravi-Moussavi A, Kheradpour P, Zhang Z, et al. (Roadmap Epigenomics Consortium) (February 2015). "Integrative analysis of 111 reference human epigenomes". Nature. 518 (7539): 317–30. Bibcode:2015Natur.518..317.. doi:10.1038/nature14248. PMC 4530010. PMID 25693563.
- Karmodiya, Krishanpal; Krebs, Arnaud R.; Oulad-Abdelghani, Mustapha; Kimura, Hiroshi; Tora, Laszlo (2012). "H3K9 and H3K14 acetylation co-occur at many gene regulatory elements, while H3K14ac marks a subset of inactive inducible promoters in mouse embryonic stem cells". BMC Genomics. 13: 424. doi:10.1186/1471-2164-13-424. PMC 3473242. PMID 22920947.
- Jurkowska, Renata Z.; Qin, Su; Kungulovski, Goran; Tempel, Wolfram; Liu, Yanli; Bashtrykov, Pavel; Stiefelmaier, Judith; Jurkowski, Tomasz P.; Kudithipudi, Srikanth; Weirich, Sara; Tamas, Raluca; Wu, Hong; Dombrovski, Ludmila; Loppnau, Peter; Reinhardt, Richard; Min, Jinrong; Jeltsch, Albert (2017). "H3K14ac is linked to methylation of H3K9 by the triple Tudor domain of SETDB1". Nature Communications. 8: 2057. Bibcode:2017NatCo...8.2057J. doi:10.1038/s41467-017-02259-9. PMID 29234025.
- "Whole-Genome Chromatin IP Sequencing (ChIP-Seq)" (PDF). Illumina. Retrieved 23 October 2019.
- "MAINE-Seq/Mnase-Seq". illumina. Retrieved 23 October 2019.
- Buenrostro, Jason D.; Wu, Beijing; Chang, Howard Y.; Greenleaf, William J. (2015). "ATAC-seq: A Method for Assaying Chromatin Accessibility Genome-Wide". Current Protocols in Molecular Biology. 109: 21.29.1–21.29.9. doi:10.1002/0471142727.mb2129s109. ISBN 9780471142720. PMC 4374986. PMID 25559105.
- Schep, Alicia N.; Buenrostro, Jason D.; Denny, Sarah K.; Schwartz, Katja; Sherlock, Gavin; Greenleaf, William J. (2015). "Structured nucleosome fingerprints enable high-resolution mapping of chromatin architecture within regulatory regions". Genome Research. 25 (11): 1757–1770. doi:10.1101/gr.192294.115. ISSN 1088-9051. PMC 4617971. PMID 26314830.
- Song, L.; Crawford, G. E. (2010). "DNase-seq: A High-Resolution Technique for Mapping Active Gene Regulatory Elements across the Genome from Mammalian Cells". Cold Spring Harbor Protocols. 2010 (2): pdb.prot5384. doi:10.1101/pdb.prot5384. ISSN 1559-6095. PMC 3627383. PMID 20150147.