Ethylenediaminediacetic acid
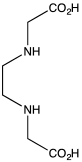
Ethylenediaminediacetic acid (EDDA) is the organic compound with the formula C2H4(NHCH2CO2H)2. It is a derivative of two molecules of glycine, wherein the amines are linked. It is a white solid.
 | |
| Names | |
|---|---|
| Other names
N,N'-ethylenediglycine | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| EC Number |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C6H12N2O4 | |
| Molar mass | 176.172 g·mol−1 |
| Appearance | white solid |
| Melting point | 228 °C (442 °F; 501 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The conjugate base is a tetradentate ligand.[1] A representative complex is Na[Co(EDDA)(CO3)].[2]
References
- Sabo, Tibor, J.; Grguric-Sipka, Sanja R.; Trifunovic, Srecko R. (2002). "Transition Metal Complexes with EDDA-Type Ligands-a Review". Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry. 32: 1661–1717. doi:10.1081/SIM-120015086.CS1 maint: uses authors parameter (link)
- Leon J. Halloran, Arlene L. Gillie, J. Ivan Legg (1978). Ethylenediamine-N,N′-Diacetic Acid Complexes of Cobalt(III). Inorg. Synth. Inorganic Syntheses. 18. pp. 103–111. doi:10.1002/9780470132494.ch17. ISBN 9780470132494.CS1 maint: uses authors parameter (link)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.