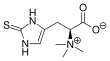
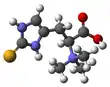
Ergothioneine
Ergothioneine is a naturally occurring amino acid and is a thiourea derivative of histidine, containing a sulfur atom on the imidazole ring.[1] This compound is made in relatively few organisms, notably Actinobacteria, Cyanobacteria, and certain fungi.[2][3] Ergothioneine was discovered in 1909 and named after the ergot fungus from which it was first purified,[4] with its structure being determined in 1911.[5]
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
3-(2-Sulfanylidene-1,3-dihydroimidazol-4-yl)-2-(trimethylazaniumyl)propanoate | |||
| Other names
L-Ergothioneine; (+)-Ergothioneine; Thiasine; Sympectothion; Ergothionine; Erythrothioneine; Thiolhistidinebetaine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.131 | ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C9H15N3O2S | |||
| Molar mass | 229.30 g/mol | ||
| Appearance | white solid | ||
| Melting point | 275 to 277 °C (527 to 531 °F; 548 to 550 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
In vitro assays, as well as in vivo animal models, are used in basic research to identify its potential biological properties.[6][7][8]
In humans, ergothioneine is acquired exclusively through the diet and accumulates in erythrocytes, bone marrow, liver, kidney, seminal fluid, and eyes.[6] Ergothioneine requires a specific transporter, ETT, also known as OCTN1 (gene symbol SLC22A4), to enter cells.[6][9] ETT expression has been confirmed in human and animal cell lines and its functional transport of ergothioneine has been observed in preliminary studies in vivo.[6][10][11] Although the effect of ergothioneine in vivo is an active area of research, its physiological role in humans is undetermined.[6]
Metabolism and sources
Ergothioneine has been found in bacteria, plants, and animals, sometimes at millimolar levels.[12] Foods found to contain ergothioneine include liver, kidney, black beans, kidney bean, and oat bran, with the highest levels in bolete and oyster mushrooms.[12][13] Levels can be variable, even within species and some tissues can contain much more than others. In the human body, the largest amounts of ergothioneine are found in erythrocytes, eye lens, semen,[5] and skin.[14]
Although many species contain ergothioneine, only a few make it; the others absorb it from their diet or, in the case of plants, from their environment.[15] Biosynthesis has been detected in Actinobacteria, such as Mycobacterium smegmatis and certain fungi, such as Neurospora crassa.[2]
The metabolic pathway to produce ergothioneine starts with the methylation of histidine to produce histidine betaine (hercynine). The sulfur atom is then incorporated from cysteine.[12][16] The biosynthetic genes of ergothioneine have been described in Mycobacterium smegmatis,[17] Neurospora crassa,[18] and Schizosaccharomyces pombe.[19]
Other species of bacteria, such as Bacillus subtilis, Escherichia coli, Proteus vulgaris, and Streptococcus, as well as fungi in the Saccharomycotina cannot make ergothioneine.[20][21]
Structure
Ergothioneine is a thiourea derivative of the betaine of histidine and contains a sulfur atom bonded to the 2-position of the imidazole ring.[8] Typical of thioureas, ergothioneine is less reactive than typical thiols such as glutathione towards alkylating agents like maleimides. It also resists oxidation by air.[12] However, ergothioneine can be slowly oxidized over several days to the disulfide form in acidic solutions.[22]
Ergothioneine derivatives
Various derivatives of ergothioneine have been reported in the literature, such as S-methyl-ergothioneine[23] or selenium-containing selenoneine.[24]
Preliminary research
Ergothioneine has antioxidant properties in vitro.[2][25] Under laboratory conditions, it scavenges hydroxyl radicals and hypochlorous acid, inhibits production of oxidants by metal ions,[26][27] and may participate in metal ion transport and regulation of metalloenzymes.[27]
Although potential effects of ergothioneine are under preliminary research, its physiological role in vivo has not been conclusively established.[6][28][29][30][31]
Safe intake levels
The Panel on Dietetic Products for the European Food Safety Authority finds that safe daily limits of 2.82 mg/kg of body weight for infants, 3.39 mg/kg for small children, and 1.31 mg/kg for adults, including pregnant and breastfeeding women.[32]
References
- "Ergothioneine". PubChem, National Center for Biotechnology Information, US National Library of Medicine. 2 November 2019. Retrieved 7 November 2019.
- Fahey RC (2001). "Novel thiols of prokaryotes". Annual Review of Microbiology. 55: 333–56. doi:10.1146/annurev.micro.55.1.333. PMID 11544359.
- Pfeiffer C, Bauer T, Surek B, Schömig E, Gründemann D (2011). "Cyanobacteria produce high levels of ergothioneine". Food Chemistry. 129 (4): 1766–1769. doi:10.1016/j.foodchem.2011.06.047.
- C. Tanret Sur une base nouvelle retiree du seigle ergote, l'ergothioneine Compt. Rend., 149 (1909), pp. 222-224
- Mann T, Leone E (January 1953). "Studies on the metabolism of semen. VIII. Ergothioneine as a normal constituent of boar seminal plasma; purification and crystallization; site of formation and function". The Biochemical Journal. 53 (1): 140–8. doi:10.1042/bj0530140. PMC 1198115. PMID 13032046.
- Cheah IK, Halliwell B (May 2012). "Ergothioneine; antioxidant potential, physiological function and role in disease". Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. 1822 (5): 784–93. doi:10.1016/j.bbadis.2011.09.017. PMID 22001064.
- Gründemann D (May 2012). "The ergothioneine transporter controls and indicates ergothioneine activity--a review". Preventive Medicine. 54 Suppl: S71-4. doi:10.1016/j.ypmed.2011.12.001. PMID 22182480.
- Hartman PE (1990). Ergothioneine as antioxidant. Meth. Enzymol. Methods in Enzymology. 186. pp. 310–8. doi:10.1016/0076-6879(90)86124-E. ISBN 978-0-12-182087-9. PMID 2172707.
- Markova NG, Karaman-Jurukovska N, Dong KK, Damaghi N, Smiles KA, Yarosh DB (April 2009). "Skin cells and tissue are capable of using L-ergothioneine as an integral component of their antioxidant defense system". Free Radical Biology & Medicine. 46 (8): 1168–76. doi:10.1016/j.freeradbiomed.2009.01.021. PMID 19439218.
- Koepsell H (2014). "Organic Cation and Zwitterion Transporters (OCTs, OCTNs)". In You G, Morris ME (eds.). Drug Transporters: Molecular Characterization and Role in Drug Disposition (Second ed.). Wiley. pp. 7–24. doi:10.1002/9781118705308.ch2. ISBN 9781118705308.
- Aruoma OI, Coles LS, Landes B, Repine JE (May 2012). "Functional benefits of ergothioneine and fruit- and vegetable-derived nutraceuticals: overview of the supplemental issue contents". Preventive Medicine. 54 Suppl: S4-8. doi:10.1016/j.ypmed.2012.04.001. PMID 22579240.
- Ey J, Schömig E, Taubert D (August 2007). "Dietary sources and antioxidant effects of ergothioneine". Journal of Agricultural and Food Chemistry. 55 (16): 6466–74. doi:10.1021/jf071328f. PMID 17616140.
- Kalač P. Edible Mushrooms. Chapter 4 - Health-Stimulating Compounds and Effects. pp 137-153. Academic Press, 2016. ISBN 9780128044551 doi:10.1016/B978-0-12-804455-1.00004-7
- Markova NG, Karaman-Jurukovska N, Dong KK, Damaghi N, Smiles KA, Yarosh DB (April 2009). "Skin cells and tissue are capable of using L-ergothioneine as an integral component of their antioxidant defense system". Free Radical Biology & Medicine. 46 (8): 1168–76. doi:10.1016/j.freeradbiomed.2009.01.021. PMID 19439218.
- Audley BS, Tan CH (1968). "The uptake of ergothioneine from the soil into the latex of Hevea brasiliensis". Phytochemistry. 7 (11): 1999–2000. doi:10.1016/S0031-9422(00)90759-3.
- Melville DB, Ludwig ML, Inamine E, Rachele JR (May 1959). "Transmethylation in the biosynthesis of ergothionelne". The Journal of Biological Chemistry. 234 (5): 1195–8. doi:10.1016/S0021-9258(18)98157-3. PMID 13654346.
- Seebeck FP (May 2010). "In vitro reconstitution of Mycobacterial ergothioneine biosynthesis". Journal of the American Chemical Society. 132 (19): 6632–3. doi:10.1021/ja101721e. PMID 20420449.
- Bello MH, Barrera-Perez V, Morin D, Epstein L (February 2012). "The Neurospora crassa mutant NcΔEgt-1 identifies an ergothioneine biosynthetic gene and demonstrates that ergothioneine enhances conidial survival and protects against peroxide toxicity during conidial germination". Fungal Genetics and Biology. 49 (2): 160–72. doi:10.1016/j.fgb.2011.12.007. PMID 22209968.
- Pluskal T, Ueno M, Yanagida M (2014). "Genetic and metabolomic dissection of the ergothioneine and selenoneine biosynthetic pathway in the fission yeast, S. pombe, and construction of an overproduction system". PLOS ONE. 9 (5): e97774. Bibcode:2014PLoSO...997774P. doi:10.1371/journal.pone.0097774. PMC 4020840. PMID 24828577.
- Genghof DS (August 1970). "Biosynthesis of ergothioneine and hercynine by fungi and Actinomycetales". Journal of Bacteriology. 103 (2): 475–8. doi:10.1128/JB.103.2.475-478.1970. PMC 248105. PMID 5432011.
- Genghof DS, Inamine E, Kovalenko V, Melville DB (November 1956). "Ergothioneine in microorganisms". The Journal of Biological Chemistry. 223 (1): 9–17. doi:10.1016/S0021-9258(18)65113-0. PMID 13376573.
- Heath H, Toennies G (February 1958). "The preparation and properties of ergothioneine disulphide". The Biochemical Journal. 68 (2): 204–10. doi:10.1042/bj0680204. PMC 1200325. PMID 13522601.
- Asmus KD, Bensasson RV, Bernier JL, Houssin R, Land EJ (April 1996). "One-electron oxidation of ergothioneine and analogues investigated by pulse radiolysis: redox reaction involving ergothioneine and vitamin C". The Biochemical Journal. 315 ( Pt 2) (2): 625–9. doi:10.1042/bj3150625. PMC 1217242. PMID 8615839.
- Yamashita Y, Yamashita M (June 2010). "Identification of a novel selenium-containing compound, selenoneine, as the predominant chemical form of organic selenium in the blood of bluefin tuna". The Journal of Biological Chemistry. 285 (24): 18134–8. doi:10.1074/jbc.C110.106377. PMC 2881734. PMID 20388714.
- Hand CE, Honek JF (February 2005). "Biological chemistry of naturally occurring thiols of microbial and marine origin". Journal of Natural Products. 68 (2): 293–308. doi:10.1021/np049685x. PMID 15730267.
- Akanmu D, Cecchini R, Aruoma OI, Halliwell B (July 1991). "The antioxidant action of ergothioneine". Archives of Biochemistry and Biophysics. 288 (1): 10–6. doi:10.1016/0003-9861(91)90158-F. PMID 1654816.
- Misiti F, Castagnola M, Zuppi C, Giardina B, Messana I (June 2001). "Role of ergothioneine on S-nitrosoglutathione catabolism". The Biochemical Journal. 356 (Pt 3): 799–804. doi:10.1042/0264-6021:3560799. PMC 1221906. PMID 11389687.
- Servillo L, D'Onofrio N, Balestrieri ML (April 2017). "Ergothioneine Antioxidant Function: From Chemistry to Cardiovascular Therapeutic Potential". Journal of Cardiovascular Pharmacology. 69 (4): 183–191. doi:10.1097/FJC.0000000000000464. PMID 28375902. S2CID 27998568.
- Halliwell B, Cheah IK, Tang RM (October 2018). "Ergothioneine - a diet-derived antioxidant with therapeutic potential". FEBS Letters. 592 (20): 3357–3366. doi:10.1002/1873-3468.13123. PMID 29851075.
- Beelman RB, Kalaras MD, Phillips AT, Richie JP (2020). "Is ergothioneine a 'longevity vitamin' limited in the American diet?". Journal of Nutritional Science. 9: e52. doi:10.1017/jns.2020.44. PMC 7681161. PMID 33244403.
- Nakamichi N, Nakao S, Nishiyama M, Takeda Y, Ishimoto T, Masuo Y, et al. (February 2020). "Oral administration of the food derived hydrophilic antioxidant ergothioneine enhances object recognition memory in mice". Current Molecular Pharmacology. 14 (2): 220–233. doi:10.2174/1874467213666200212102710. PMID 32048982.
- Turck D, Bresson JL, Burlingame B, Dean T, Fairweather-Tait S, Heinonen M, et al. (November 2017). "Statement on the safety of synthetic l-ergothioneine as a novel food - supplementary dietary exposure and safety assessment for infants and young children, pregnant and breastfeeding women". EFSA Journal. European Food Safety Authority. 15 (11): e05060. doi:10.2903/j.efsa.2017.5060. PMC 7010164. PMID 32625352.
External links
- Ergothioneine Human Metabolome Database
- Ergothioneine KEGG Compound
- Ergothioneine Tetrahedron