CYC1
Cytochrome c1, heme protein, mitochondrial (CYC1), also known as UQCR4, MC3DN6, Complex III subunit 4, Cytochrome b-c1 complex subunit 4, or Ubiquinol-cytochrome-c reductase complex cytochrome c1 subunit is a protein that in humans is encoded by the CYC1 gene. CYC1 is a respiratory subunit of Ubiquinol Cytochrome c Reductase (complex III), which is located in the inner mitochondrial membrane and is part of the electron transport chain. Mutations in this gene may cause mitochondrial complex III deficiency, nuclear, type 6.[5][6][7]
Structure
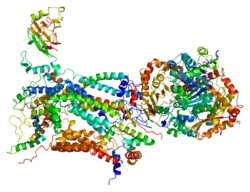
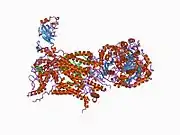
CYC1 is located on the q arm of chromosome 8 in position 24.3 and has 8 exons.[5] The CYC1 gene produces a 13.5 kDa protein composed of 130 amino acids.[8][9] CYC1 belongs to the cytochrome c family. CYC1 is a phosphoprotein and subunit of Ubiquinol Cytochrome c Reductase that binds heme groups. It has helix, transit peptide, and transmembrane domains and contains 9 alpha helixes, 5 beta strands, and 3 turns. The transmembrane protein passes through the inner mitochondrial membrane once and the majority of the protein is found on the intermembrane side. CYC1 contains covalent heme bindings sites at positions 121 and 124 and heme axial ligand iron-metal binding sites at positions 125 and 244.[6][7]
Function
CYC1 encodes a protein that is located in the inner mitochondrial membrane and is part of Ubiquinol Cytochrome c Reductase (complex III). The encoded protein, CYC1, is a respiratory subunit of the cytochrome bc1 complex, which plays an important role in the mitochondrial respiratory chain by transferring electrons from the Rieske iron-sulfur protein to cytochrome c.[5][6][7]
Species
CYC1 is a human gene that is conserved in chimpanzee, Rhesus monkey, dog, cow, mouse, rat, zebrafish, fruit fly, mosquito, C. elegans, S. cerevisiae, K. lactis, E. gossypii, S. pombe, N. crassa, A. thaliana, rice, and frog.[10] There are orthologs of CYC1 in 137 known organisms.[11]
Clinical Significance
Variants of CYC1 have been associated with mitochondrial complex III deficiency, nuclear, type 6. Mitochondrial complex III deficiency, nuclear, type 6 is an autosomal recessive disorder of the mitochondrial respiratory chain resulting from a defect in Ubiquinol Cytochrome c Reductase (complex III) that leads to reduced complex III activity. Clinical features tend to emerge in early childhood and include episodic acute lactic acidosis, ketoacidosis, insulin-responsive hyperglycemia, liver dysfunction, encephalopathy, and associated infection, although psychomotor development may remain normal. Pathogenic mutations have included c.288G>T, p.Trp96Cys and c.643C>T p. Leu215Phe.[6][7][12]
Interactions
CYC1 has 78 protein-protein interactions with 72 of them being co-complex interactions.[13] CYC1 is one of 11 subunits of Ubiquinol Cytochrome c Reductase (b1-c complex) that includes the respiratory subunits cytochrome b, cytochrome c1 (CYC1), UQCRFS1, the core proteins UQCRC1 and UQCRC2, and the low-molecular weight proteins UQCRH, UQCRB, UQCRQ, UQCR10, UQCR11, as well as an additional cleavage product of UQCRFS1.[6][7] Additionally, CCP1, CDKA-1, and CDKB1-1 have also been found to interact with CYC1.[13]
References
- GRCh38: Ensembl release 89: ENSG00000179091 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000022551 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Entrez Gene: CYC1 cytochrome c-1".
- "CYC1 - Cytochrome c1, heme protein, mitochondrial precursor - Homo sapiens (Human) - CYC1 gene & protein". www.uniprot.org. Retrieved 2018-07-31.
- "UniProt: the universal protein knowledgebase". Nucleic Acids Research. 45 (D1): D158–D169. January 2017. doi:10.1093/nar/gkw1099. PMC 5210571. PMID 27899622.
- Yao, Daniel. "Cardiac Organellar Protein Atlas Knowledgebase (COPaKB) —— Protein Information". amino.heartproteome.org. Archived from the original on 2018-08-01. Retrieved 2018-07-31.
- Zong NC, Li H, Li H, Lam MP, Jimenez RC, Kim CS, Deng N, Kim AK, Choi JH, Zelaya I, Liem D, Meyer D, Odeberg J, Fang C, Lu HJ, Xu T, Weiss J, Duan H, Uhlen M, Yates JR, Apweiler R, Ge J, Hermjakob H, Ping P (October 2013). "Integration of cardiac proteome biology and medicine by a specialized knowledgebase". Circulation Research. 113 (9): 1043–53. doi:10.1161/CIRCRESAHA.113.301151. PMC 4076475. PMID 23965338.
- "CYC1 cytochrome c1 [Homo sapiens (human)]". National Center for Biotechnology Information. U.S. National Library of Medicine. Retrieved 2016-07-29.
- "ortholog_gene_1537[group]". National Center for Biotechnology Information. U.S. National Library of Medicine. Retrieved 2016-07-29.
- Gaignard P, Menezes M, Schiff M, Bayot A, Rak M, Ogier de Baulny H, Su CH, Gilleron M, Lombes A, Abida H, Tzagoloff A, Riley L, Cooper ST, Mina K, Sivadorai P, Davis MR, Allcock RJ, Kresoje N, Laing NG, Thorburn DR, Slama A, Christodoulou J, Rustin P (August 2013). "Mutations in CYC1, encoding cytochrome c1 subunit of respiratory chain complex III, cause insulin-responsive hyperglycemia". American Journal of Human Genetics. 93 (2): 384–9. doi:10.1016/j.ajhg.2013.06.015. PMC 3738829. PMID 23910460.
- "81 binary interactions found for search term CYC1". IntAct Molecular Interaction Database. EMBL-EBI. Retrieved 2018-08-25.<
External links
- Human CYC1 genome location and CYC1 gene details page in the UCSC Genome Browser.
Further reading
- Moon HS, Yang JS (February 2006). "Role of HIV Vpr as a regulator of apoptosis and an effector on bystander cells". Molecules and Cells. 21 (1): 7–20. PMID 16511342.
- Suzuki H, Hosokawa Y, Nishikimi M, Ozawa T (January 1989). "Structural organization of the human mitochondrial cytochrome c1 gene". The Journal of Biological Chemistry. 264 (3): 1368–74. PMID 2536365.
- Nishikimi M, Ohta S, Suzuki H, Tanaka T, Kikkawa F, Tanaka M, Kagawa Y, Ozawa T (April 1988). "Nucleotide sequence of a cDNA encoding the precursor to human cytochrome c1". Nucleic Acids Research. 16 (8): 3577. doi:10.1093/nar/16.8.3577. PMC 336517. PMID 2836796.
- Tanaka Y, Ashikari T, Shibano Y, Amachi T, Yoshizumi H, Matsubara H (June 1988). "Construction of a human cytochrome c gene and its functional expression in Saccharomyces cerevisiae". Journal of Biochemistry. 103 (6): 954–61. doi:10.1093/oxfordjournals.jbchem.a122393. PMID 2844747.
- Shimomura Y, Nishikimi M, Ozawa T (December 1985). "Novel purification of cytochrome c1 from mitochondrial Complex III. Reconstitution of antimycin-insensitive electron transfer with the iron-sulfur protein and cytochrome c1". The Journal of Biological Chemistry. 260 (28): 15075–80. PMID 2999105.
- Nishikimi M, Suzuki H, Ohta S, Sakurai T, Shimomura Y, Tanaka M, Kagawa Y, Ozawa T (May 1987). "Isolation of a cDNA clone for human cytochrome c1 from a lambda gt11 expression library". Biochemical and Biophysical Research Communications. 145 (1): 34–9. doi:10.1016/0006-291X(87)91283-6. PMID 3036122.
- Smith HT, Ahmed AJ, Millett F (May 1981). "Electrostatic interaction of cytochrome c with cytochrome c1 and cytochrome oxidase". The Journal of Biological Chemistry. 256 (10): 4984–90. PMID 6262312.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Duncan AM, Ozawa T, Suzuki H, Rozen R (January 1994). "Assignment of the gene for the cytochrome c1 subunit of the mitochondrial cytochrome bc1 complex (CYC1) to human chromosome 8q24.3". Genomics. 19 (2): 400–1. doi:10.1006/geno.1994.1084. PMID 8188279.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Valnot I, Kassis J, Chretien D, de Lonlay P, Parfait B, Munnich A, Kachaner J, Rustin P, Rötig A (June 1999). "A mitochondrial cytochrome b mutation but no mutations of nuclearly encoded subunits in ubiquinol cytochrome c reductase (complex III) deficiency". Human Genetics. 104 (6): 460–6. doi:10.1007/s004390050988. PMID 10453733. S2CID 30584267.
- Jacotot E, Ferri KF, El Hamel C, Brenner C, Druillennec S, Hoebeke J, Rustin P, Métivier D, Lenoir C, Geuskens M, Vieira HL, Loeffler M, Belzacq AS, Briand JP, Zamzami N, Edelman L, Xie ZH, Reed JC, Roques BP, Kroemer G (February 2001). "Control of mitochondrial membrane permeabilization by adenine nucleotide translocator interacting with HIV-1 viral protein rR and Bcl-2". The Journal of Experimental Medicine. 193 (4): 509–19. doi:10.1084/jem.193.4.509. PMC 2195906. PMID 11181702.
- Tafani M, Karpinich NO, Hurster KA, Pastorino JG, Schneider T, Russo MA, Farber JL (March 2002). "Cytochrome c release upon Fas receptor activation depends on translocation of full-length bid and the induction of the mitochondrial permeability transition". The Journal of Biological Chemistry. 277 (12): 10073–82. doi:10.1074/jbc.M111350200. PMID 11790791.
- Muthumani K, Hwang DS, Desai BM, Zhang D, Dayes N, Green DR, Weiner DB (October 2002). "HIV-1 Vpr induces apoptosis through caspase 9 in T cells and peripheral blood mononuclear cells". The Journal of Biological Chemistry. 277 (40): 37820–31. doi:10.1074/jbc.M205313200. PMID 12095993.
- Muthumani K, Zhang D, Hwang DS, Kudchodkar S, Dayes NS, Desai BM, Malik AS, Yang JS, Chattergoon MA, Maguire HC, Weiner DB (July 2002). "Adenovirus encoding HIV-1 Vpr activates caspase 9 and induces apoptotic cell death in both p53 positive and negative human tumor cell lines". Oncogene. 21 (30): 4613–25. doi:10.1038/sj.onc.1205549. PMID 12096338.
- Brenner C, Kroemer G (May 2003). "The mitochondriotoxic domain of Vpr determines HIV-1 virulence". The Journal of Clinical Investigation. 111 (10): 1455–7. doi:10.1172/JCI18609. PMC 155055. PMID 12750393.
- Lum JJ, Cohen OJ, Nie Z, Weaver JG, Gomez TS, Yao XJ, Lynch D, Pilon AA, Hawley N, Kim JE, Chen Z, Montpetit M, Sanchez-Dardon J, Cohen EA, Badley AD (May 2003). "Vpr R77Q is associated with long-term nonprogressive HIV infection and impaired induction of apoptosis". The Journal of Clinical Investigation. 111 (10): 1547–54. doi:10.1172/JCI16233. PMC 155040. PMID 12750404.
- Yuan J, Murrell GA, Trickett A, Wang MX (June 2003). "Involvement of cytochrome c release and caspase-3 activation in the oxidative stress-induced apoptosis in human tendon fibroblasts". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1641 (1): 35–41. doi:10.1016/S0167-4889(03)00047-8. PMID 12788227.
- An J, Chen Y, Huang Z (April 2004). "Critical upstream signals of cytochrome C release induced by a novel Bcl-2 inhibitor". The Journal of Biological Chemistry. 279 (18): 19133–40. doi:10.1074/jbc.M400295200. PMID 14966123.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.